LONG-TERM ECOLOGICAL RESEARCH (LTER)
IN LAND/OCEAN MARGIN ECOSYSTEMS
Plum Island Sound Comparative Ecosystem Study (PISCES):
Effects of changing land cover, climate, and sea level
on estuarine trophic dynamics
Charles S. Hopkinson, Jr.1
Dennis Allen2
Robert Buchsbaum3
Linda Deegan1
Michelle Dionne4
Anne Giblin1
John Hobbie1
James Morris5
Bruce Peterson1
Joseph Vallino1
Charles Vorosmarty6
1Ecosystems Center
Marine Biological Laboratory
Woods Hole, MA
2University of South Carolina
Georgetown, SC
3Massachusetts Audubon Society
Wenham, MA
4Wells National Estuarine Research Reserve
Wells, ME
5University of South Carolina
Columbia, SC
6University of New Hampshire
Durham, NH
June 1997
PROJECT SUMMARY
Human activities in rivers and watersheds have altered enormously the timing, magnitude and nature of inputs of materials such as water, sediments, nutrients and organic matter to estuaries. An important but neglected linkage between land and coastal waters is the input of dissolved and particulate organic carbon and organic nitrogen. This long-term ecological research (LTER) in land/ocean margin ecosystems will focus on the following question and hypotheses:
How will trophic structure and primary and secondary productivity in estuaries be affected by changes in organic matter, nutrient and water fluxes cause by changing land cover, climate and sea level?
Hypothesis 2. The variability in land, ocean and atmospheric forcing is a key component determining the fate of allochthonous and autochthonous materials and the location and magnitude of primary and secondary production.
The study builds on the existing long-term research record at Plum Island Sound, MA, and at the North Inlet, SC and Wells, ME National Estuarine Research Reserves. Intensive work will take place at Plum Island Sound, and specific comparative studies at North Inlet and Wells. The research is designed to take advantage of natural differences between these systems to determine the importance of inorganic and organic matter interactions in controlling trophic structure, production and efficiency.
The research advances ecological science in a number of ways. The project addresses the question: is there a coherent response of ecosystems in an entire region to a large-scale disturbance? Here the region is the East Coast of the U.S. and the disturbance is long-term sea level rise. The project will also ask: how do the biota of different biogeographic provinces respond to similar disturbances in nutrient inputs, freshwater supply, and organic matter availability?
The project will make a unique contribution to our understanding of
land-margin ecosystems by determining how changing nutrient supply interacts
with the quality and quantity of organic matter to affect estuarine trophic
structure. It will also develop modeling method to link highly variable
physical drivers, here tides, long-term sea level rise, river inflow, and
water circulation, with the understanding of ecological processes. The
dynamic models developed will be useful in a variety of ways for predicting
effects of various coastal management options. For example, the spatially-explicit
models to be developed can be used to predict the effects of changing land
use on eutrophication and fish production.

RESULTS OF PRIOR RESEARCH
NSF Grant #OCE-9214461: Hopkinson, Deegan, Fry, Giblin, Hobbie, Peterson, Finn and Buchsbaum. $1,600,000. Sept 1992-1996 with 1 yr extension. The Plum Island Sound Comparative Ecosystem Study (PISCES), an LMER project, addresses two hypotheses concerning the importance to estuarine ecosystems of organic matter inputs from watersheds with various land covers and the interaction between organic matter and inorganic nutrients in controlling estuarine food web structure, efficiency and productivity. Research on this 4 year project has focused on: the watersheds, microbial utilization and remineralization of organic matter, effects of organic matter - nutrient interactions on trophic structure and production, macrofaunal community structure, and patterns of benthic, pelagic and whole system production and respiration. Several simulation models were developed to guide, summarize and synthesize research. Considerable baseline, descriptive data was collected.
Scientific Infrastructure: Results have been presented at many local, national and international scientific and lay meetings and symposia. Over 43 publications were printed, accepted for publication or submitted to peer-reviewed journals (shown with "*" in Bib). Participation of undergraduate (10), graduate (5), international (2) and post-doctoral scholars (3) has contributed to the success of our program. Seven of our REU students published their research in the peer-reviewed literature. Site bibliography and considerable data are on our web page (http://www.mbl.edu/plumisla.html).
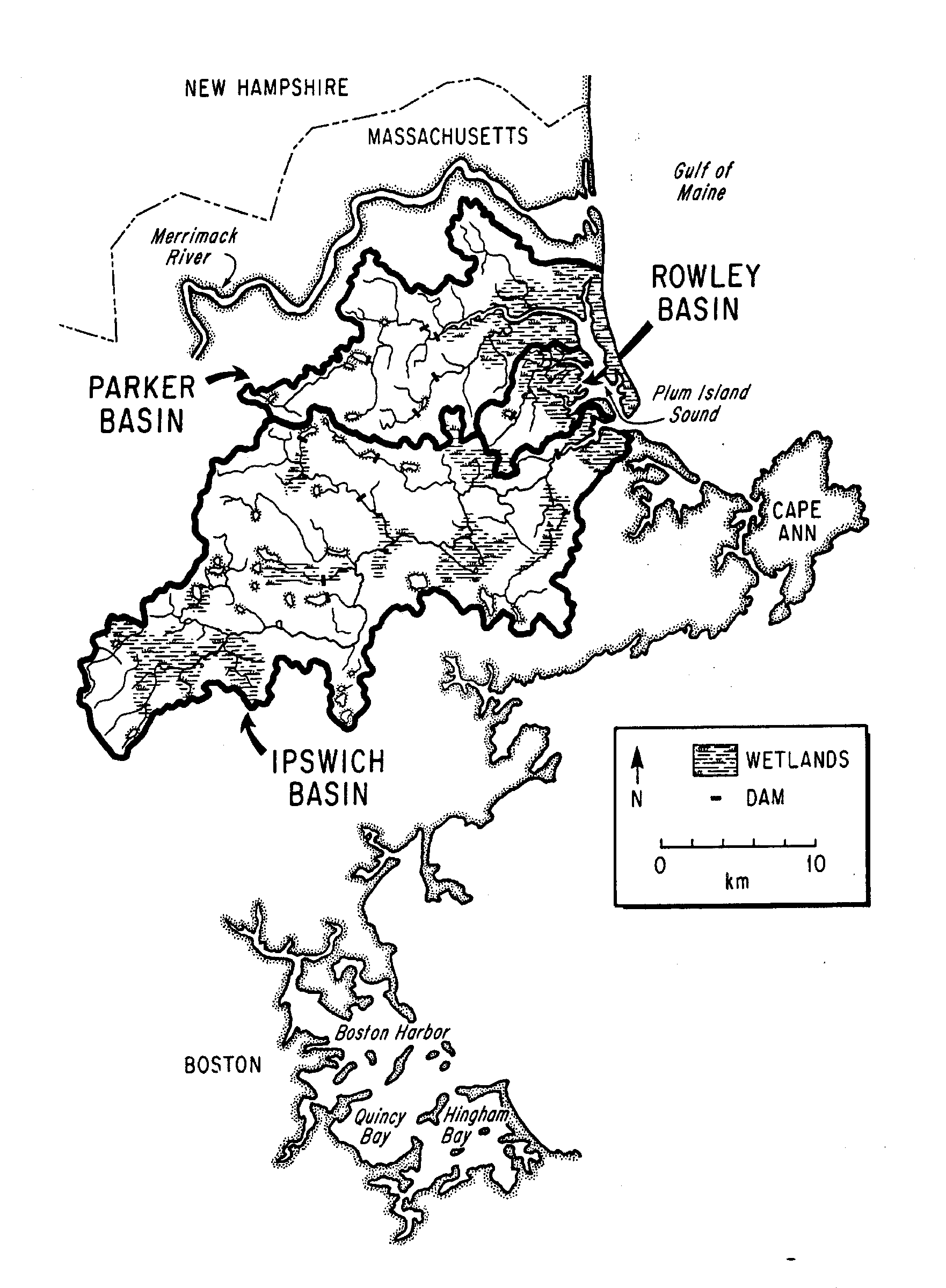
Watershed: Keynote- Major upland NO3- loads, primarily from urban areas, are retained with 80-90% efficiency in rivers resulting in DON being the major form of N export to estuary. Land use, economic activity and population are changing rapidly in the three river basins comprising the 585 km2 upland watershed of the Plum Island Sound Estuary. We have 1) characterized historical landuse for the Parker, Rowley and Ipswich River watersheds, 2) developed digital databases (soils, land cover, slope, streams, erosivity, etc.) for use in hydrologic modeling, 3) characterized runoff chemistry for several small catchments and 4) modeled water, organic matter and nutrient discharge to the estuary (Finn and Hopkinson, 1997).
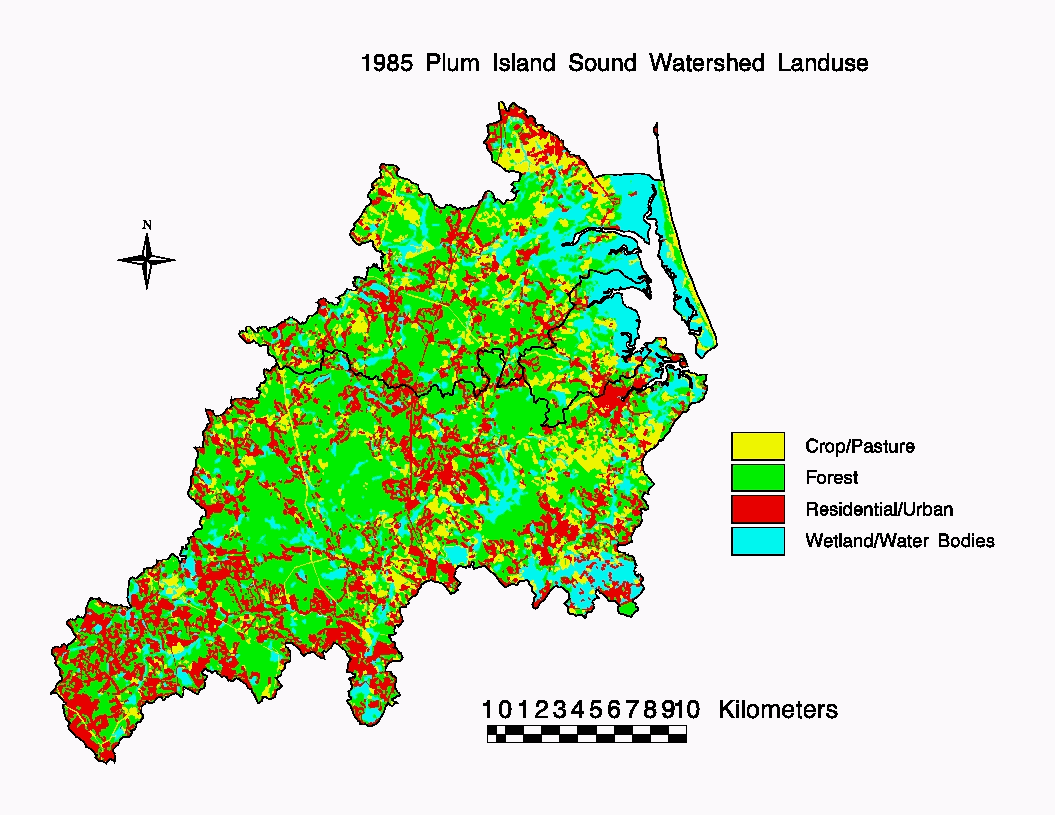
Land cover (Fig 1) in the watershed in 1985 was
»
50% forest, 25% urban/suburban, 13% wetland, and 12% agricultural. During
the past 25 years urban area increased substantially, largely at the expense
of agricultural (down 50%) and forested land (down 8%).
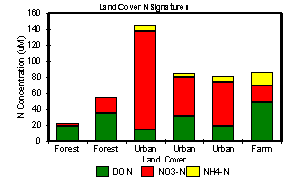
Runoff characteristics and streamwater chemistry (spm, C, N and P) of streams draining various land covers differed substantially. Total N was dominated by the dissolved fractions for all land covers (Fig 2). Total dissolved N (TDN) concentration was highest in urban runoff and least in forest runoff. While TDN was dominated by inorganic N (primarily NO3-) in urban runoff, dissolved organic N (DON) comprised up to 90% of the total in forested streams. Agricultural runoff was intermediate in concentration, with inorganic and organic fractions of roughly equal concentration.
Material loading and export budgets were created by scaling subcatchment results to entire watersheds using a combination of hydrologic models (TR-55, Brook6, and GWLF). Our riverine budgets did not balance. For N we estimated 515-975 Mg N yr-1 entered the river, while only 91-106 Mg yr-1 were discharged to the estuary. Thus in stream processing removed 80-90% of inputs from land probably through a combination of processes including vegetation uptake, sedimentation, organic matter accumulation and burial and denitrification. DON was the major form of N exported from the watershed.
Estuarine Hydrodynamics: Keynote- Residence time varies spatially and temporally from as long as 34 days in upper estuary to as little as 0.5 d in lower estuary depending on river flow.Salinity distributions, dye release studies and parameter estimation techniques were used to determine longitudinal dispersion coefficients in the Estuary. Two hydrodynamic models were developed: 1) a 1-D tidally averaged, advection-dispersion model (Vallino and Hopkinson in press) and 2) a 1-D advection-dispersion model that accounts for marsh flooding (Vorosmarty and Loder 1994). These models were used to investigate characteristic mixing-time scales, average age, residence time and transit time, and to contrast nutrient dynamics during spring and neap tides. These time scales vary from hours to 2 months depending on location and freshwater discharge. Transport in the upper estuary can be either advection or dispersion dominated depending on discharge, while the lower estuary is always dominated by dispersive terms.
Microbial Utilization and Composition of Watershed-derived Organic Matter: Keynotes - Novel bioenergetic model replaces Monod growth kinetics; Organic matter from various land covers differs in chemical characteristics, ability to support bacterial growth, and potential for N immobilization. Effort focused on chemically characterizing organic matter from the variety of estuarine sources including plankton, macrophytes and runoff from several land covers. Bioassays were used to measure organic matter lability, patterns of nutrient immobilization or remineralization and bacterial growth rates and efficiency (Normann et al. 1995, Hullar et al. 1996). This work is the cornerstone of our research on organic matter - nutrient interaction effects on food web structure and efficiency.
Bioenergetic Model: A bioenergetic model was developed to examine growth dynamics associated with bacterial utilization of DOM, NH4+ and NO3- (Vallino et al. 1996). A novel, optimization approach was taken that provides more information on bacterial growth kinetics than Monod-type models that are typically employed to describe bacterial growth. The model provides a means to predict bacterial carbon yield, growth rate and N processing and is a bridge between chemical and biological characterization of DOM: bacterial dynamics are predicted from DOM chemical characteristics. Model results demonstrate that bacterial growth can not always be explained by a single constraint (such C:N ratio of substrate) as bacteria allocate resources to maximize growth rate subject to kinetic, thermodynamic and mass balance constraints. The model suggests that bacterial growth and yield can be predicted from the degree of reduction of the substrate (y ). We have been devoting considerable time applying the model to field situations and attempting to differentially characterize labile and recalcitrant pools of bulk DOM in natural waters.
Bioassays: Globally, changes in watershed landuse
have increased levels of organic carbon transported to the sea by 3-5 times
over natural levels. Yet it is unclear how much of the DOM entering estuaries
is actually used by microbes and contributes to the food web and how much
resists degradation and is exported offshore. To better understand this,
we have taken advantage of "natural" differences in DOM composition and
conducted experiments, during high and low flow periods, with runoff water
from urban, agricultural and forested land covers (Uhlenhopp et al. 1995,
Hobbie et al. 1996). DOM from these sites ranged in concentration from
310 to 870 µM C, C/N from 10 to 40, and DIN from <10 to > 50 µM
N. Lability was measured with bacterial growth bioassays where bacterial
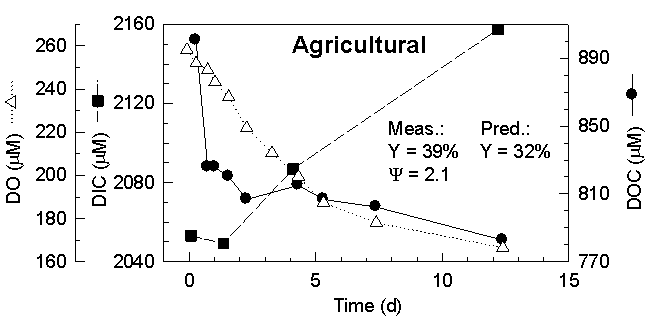
#s, DO, DIC, DOC, DON and nutrients were monitored over time. Results (Fig
3) showed that labile DOC pools ranged from ~40 µM C in the forest
to ~100 µM C in the agricultural runoff and did not vary much over
the course of a year. The percent of the bulk pool that was labile was
small, similar between sites (5-15%) and varied little over time. Bacterial
yield during the first 3 d was compared to the bacterial yield predicted
by the bioenergetic model based on the estimated degree of reduction of
the DOM. Predicted bacterial yield (29, 32 and 43%) was in good agreement
with that observed (29, 39, 52%: Fig 3). We further observed that
N was remineralized during low flow when DIN was generally low and immobilized
during high flow when DIN was high.
LMER DOM Lability Intercomparison: To assess the generality of DOM quality - bacterial growth relations identified in our Plum Island studies, we coordinated an LMER intercomparison of riverine DOM chemical characteristics and bioavailability (our site, Columbia River, Satilla River, Susquehanna River, Childs River). Large differences in chemical composition and bacterial growth were observed across systems (Hopkinson et al. submitted). Chemical characteristics (aliphatic C content and degree of substrate reduction) suggested similar patterns of lability between sites in the order Columbia > Susquehanna > Parker > Childs > Satilla. Bacterial growth rates followed this pattern, ranging from 1.3-1.7 ug d-1 mg-1 DOC. Bioassays confirmed the bioenergetic model relation between substrate reduction and bacterial growth (Vallino et al. 1996).
Organic Matter - Nutrient Interaction Effects on Higher Trophic Levels: Keynotes - Mesocosm experiments support trophic model analysis of the effect of organic matter on foodweb efficiency; stable isotope analysis of food sources and consumers indicates preferential utilization of low quality marsh detritus by benthivores.
Mesocosm Experiments: We conducted experiments
in 10 m3 plastic bags to examine differential effects of phytoplankton
derived organic C vs terrestrial organic C on planktonic community structure
and production. To 4 mesocosms with natural, intact planktonic communities
(through larval Menidia fish) were added either: A) control - nothing,
B)
DOM from dead leaf litter leachate: final DOC up from ~100 to 500 µM,
C)
daily additions of inorganic N, P and Si, and D) leaf leachate DOM
as in (B) plus daily additions of nutrients as in (C). DI13C
was added to each bag to trace phytoplankton derived C through the food
web. Dramatic differences in community structure and production resulted.
Gross production and net community production were highest in the nutrient
enriched (C) treatment and lowest in the litter leachate (B) treatment.
Whereas primary production was dominated by diatoms > 20 µm in the
nutrient enriched bag, 85-90% of production was attributed to phytoplankton
< 20 µm in the control (A) and DOM (B) bags. Food web efficiency
was low in the DOM amendment bags (B&D) with 80% of respiration attributable
to organisms < 1 µm and only 20% attributable to organisms > 20
µm. In contrast, trophic transfer was substantially greater in the
nutrient addition bag as evidenced by ~50% of system respiration being
attributable to organisms between 20-80 µM and >80 µm in size.
Less than 50% of total respiration was attributable to bacteria. Species
composition changed with treatments as well. In the DOM bag (B), Podon
and Parvocalanus became the dominant zooplankton while in the DIN bag (C),
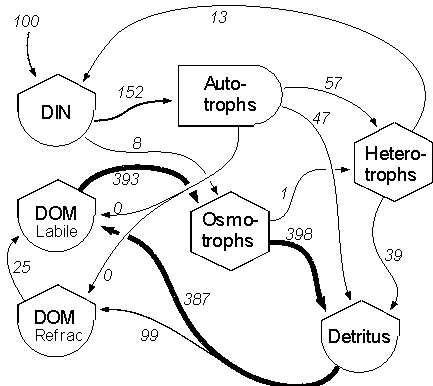
Acartia quickly became dominant. A N-flow model for bag (D) illustrates
the dominance of the microbial loop and the trivial transfer of N to higher
trophic levels (Fig 4). These results confirm previous flow model
predictions about the effects of organic matter vs nutrients on trophic
structure and production (Deegan et al. 1996): inorganic nutrients support
a short, efficient and productive food web while low quality organic matter
without inorganic nutrients support an inefficient, microbe-dominated food
web with low levels of small phytoplankton production.
Sources of Organic Matter Supporting Higher Trophic Levels in the Estuary: The importance of phytoplankton, benthic microalgae, marsh grass and terrestrial organic matter to the estuarine food web was evaluated using stable isotopes of C, N and S (Deegan and Garritt 1997). Strong spatial differences in relative importance of organic matter sources existed within the estuary. While there is substantial spatial heterogeneity in organic matter sources within the estuary, consumers utilize sources produced in the same region. Terrestrial organic matter was not evident in the food webs of the middle and lower estuary and its importance in the oligohaline region was equivocal as there was not a distinct separation in isotopic signatures between fresh marsh grass and terrestrial organic matter. There was an interesting dichotomy in the relative importance of phytoplankton vs marsh detritus in the diets of pelagic vs benthic organisms. Benthic consumers reflected a greater dependence on marsh detritus.
LMER Intercomparison of Organic Matter Sources Fueling Higher Trophic Levels: We have been coordinating an LMER intercomparison of the relative importance to higher trophic levels of the dominant organic matter sources to estuaries: terrestrial organic matter, algae and macrophytes. There are large differences in the relative importance of these types of organic matter across systems. Stable isotopes are being used to evaluate the relative importance of the various organic matter sources to key functional groups of the food web. A goal is to determine a discount factor for the various organic matter sources that indicates the preferential utilization by functional groups of one source over another relative to the loading. This intercomparison effort is ongoing.
Macrofaunal Community Structure: Keynotes - Low number of fish species in estuary reflects nature of Acadian Biogeographic Province; Six-fold increase in forage fish density between 1965 and 93/4. The current status of the estuary as a habitat for important marine fish and shellfish was ascertained using seine and trawl samplings (Buchsbaum 1996). Sampling sites and techniques were similar to those of earlier surveys conducted by the Massachusetts Division of Marine Fisheries (Jerome et al. 1967). The distribution of organisms varied in the different habitats of the estuary. In open water and sandy substrata, Menidia, Fundulus and Crangon dominated. In muddy salt marsh habitats the same species were present but Fundulus was the dominant and Palaemonetes was also common. In brackish, riverine habitats Morone, Alosa and Rhithropanopeus were the most common organisms. In comparison to the 1965 survey, the average density of fish was about 6-fold higher in 1993-94. In both studies Fundulus and Menidia were dominants in the community but species richness based on seine sampling was slightly higher in 1993-4. Explanations for the differences in density between years is not obvious, but may be related to marked differences in average sea level or river discharge during critical periods in the development of these organisms. Long term data are needed to adequately explain whether the differences between 1965 and 1993/4 are caused by regional and environmental factors or whether fish populations naturally exhibit short term variability.
Nutrient Cycling and Metabolism: Keynotes
- Temporal variability in tidal amplitude and river discharge affect spatial
patterns of nutrient cycling, benthic nutrient fluxes and primary production;
Estuarine waters are heterotrophic and therefore net sources of CO2
and inorganic nutrients. Temporal and spatial
aspects of metabolism and nutrient cycling were documented, including primary
production (Alderman et al. 1995, Balsis et al. 1995), spring-neap tidal
contrasts in marsh-water organic matter and nutrient exchange (Vorosmarty
and Loder 1994), benthic metabolism and nutrient exchange (Hopkinson et
al. submitted), and inorganic nutrient and dissolved organic matter dynamics.
High primary production occurs in the lower estuary during winter in association
with the Gulf of Maine spring bloom. Production is high during summer in
the oligohaline region but only when mixing time scales are long relative
to phytoplankton turnover time. The benthos is a major site of organic
matter degradation and because of denitrification and the high C/N nature
of organic matter processed in the benthos, it exerts a net heterotrophic
influence on the system (ie, C production from recycled N << organic
carbon remineralized). Seasonal and annual variations in porewater salinity
of oligohaline and brackish sediments alters the timing of NH4+
release/storage in sediments. The overall system is heterotrophic with
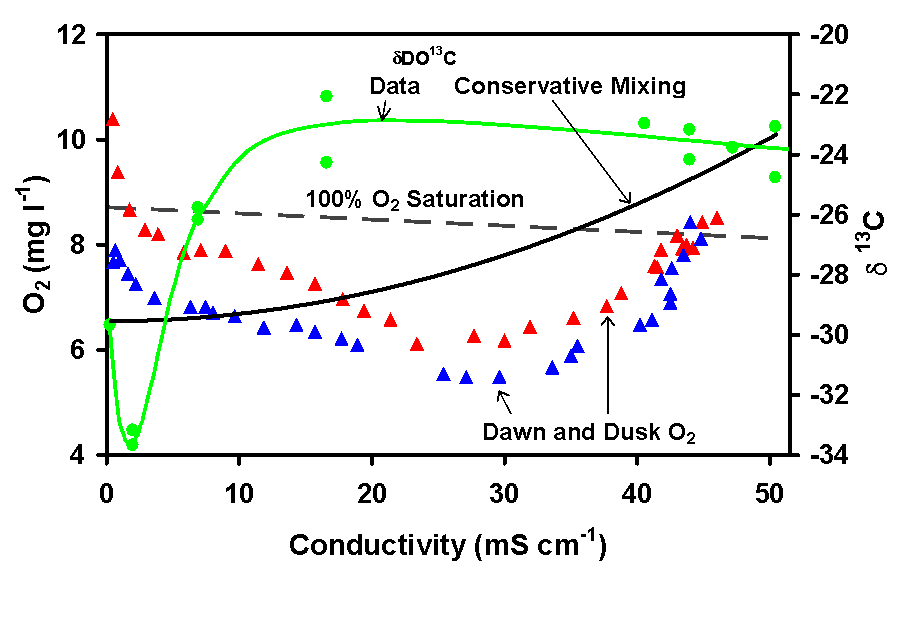
DO often depressed to 60% of saturation in mid-estuary (Fig 5).
The region of highest heterotrophy coincides with organic matter inputs
from marshes as evidenced by dDO13C
gradients in the water column (Fig. 5).
Whole System Synthesis: Keynote
- Generalized estuarine metabolism model evaluates patterns of production,
nutrient cycling and autotrophy/heterotrophy caused by variations in river
discharge and material loading from land. Flow
model analysis and simulation models have been used from the outset of
our research program to guide research, help in designing experiments,
synthesize results of microcosm and mesocosm experiments and to extend
experimental and process based research results to the field. In addition
to models described above, we have used flow model analysis to understand
the effects of organic matter - nutrient interactions on estuarine trophic
dynamics (Deegan et al. 1995). We developed a generalized estuarine metabolism
model to explore the effects of land use change and river modifications
on patterns of production, respiration and nutrient cycling (Hopkinson
and Vallino 1995). The model was useful for evaluating the effect of residence
time, organic matter quality, and DON:DIN and POM:DOM loading ratios on
patterns of net ecosystem metabolism.
INTRODUCTION
Human activities in rivers and watersheds have altered enormously the timing, magnitude and nature of inputs of materials such as water, sediments, nutrients and organic matter to estuaries (Hopkinson and Vallino 1995). One of the dominant themes of estuarine research in recent decades has been the effects of inorganic nutrients on eutrophication of coastal waters. Studies have detailed how inorganic nutrient inputs from land influence primary productivity, the depletion of oxygen, habitats, and trophic relationships in coastal waters. However, another important but neglected linkage between land and coastal waters is the input of dissolved and particulate organic carbon and organic nitrogen. Organic nitrogen inputs are frequently greater than inorganic nitrogen inputs and in many cases the input of organic carbon from land is greater than the in situ production of organic carbon. Despite the quantitative importance of organic matter inputs from land, there are few data on its actual use in coastal waters and even fewer on effects of organic matter loading on coastal ecosystem trophic structure and trophic productivity. Yet, in other ecosystems such as rivers, lakes, and the deep-sea, effects of allochthonous organic matter inputs on trophic structure and ecosystem metabolism have been well documented.
Despite an awareness of large scale, long-term changes in watersheds, we do not fully understand the consequences of activities such as damming of rivers, land use conversion, and removal of floodplains. Often human influences on river systems are opposing. For instance land clearing increases drainage basin sediment yield while damming decreases sediment discharge.
Climate variability and long-term patterns of climate change also can have immense effects on the timing, magnitude and nature of material inputs. Infrequent storms can accomplish in days what normally occurs over decades. For instance, heavy rainfall associated with tropical storm Agnes increased Susquehanna River flow to record levels, which resulted in 30 years of "normal" sediment discharge occurring in one week (Hirshberg and Schubel 1979).
Variations and long-term increases in sea level affect estuaries from their seaward end. Tides and sea level have significant effects on water and marsh sediment salinity, plant community composition (Olff et al. 1988), primary production (Morris and Haskin 1990, Morris 1995), access to marsh surface habitats by nekton (Rozas 1995) and sediment accretion (Callaway et al. 1996).
The interaction of organic matter and nutrient inputs from land and variations in the external forcings (climate, land use, river discharge, sea level) regulating estuarine mixing and residence time will dictate the extent of nutrient and organic matter processing during estuarine transport and will determine the spatial patterns of productivity and trophic structure. Our long-term research program will focus on the following question and hypotheses:
How will trophic structure and primary and secondary productivity in estuaries be affected by changes in organic matter, nutrient and water fluxes caused by changing land cover, climate and sea level?
Hypothesis 2. The variability in land, ocean and atmospheric forcing is a key component determining the fate of allochthonous and autochthonous materials and the location and magnitude of primary and secondary production.
The proposed study will consist of 1) site-specific research
at Plum Island Sound, and 2) comparative ecosystem research at North Inlet,
SC and Wells, ME national estuarine research reserves. Plum Island Sound
is in the cold-water Acadian Biogeographic Province (Fig 6) and
lies at the interface of a thin soiled, formerly glaciated New England
watersheds and the highly productive Gulf of Maine. Three watersheds with
differing levels and rates of development drain into the Plum Island Sound
tidal marsh estuary providing a wide range in the quantity and quality
of inorganic and organic matter loading. In addition experimental manipulations,
our research program is designed to take advantage of these natural experiments
within and between estuaries to determine the importance of inorganic and
organic matter interactions in controlling trophic structure, trophic production
and trophic efficiency.
Justification - Hypothesis 1. Organic Matter - Nutrient Interactions and Trophic Structure: The input of organic matter from land to coastal waters is substantial. Over the entire earth, 1-2% of terrestrial NPP (net primary production) or 1-14 gC m-2yr-1 (Meybeck 1982) is exported to estuaries; this is equivalent to 28% of estuarine NPP (Wollast and Billen 1981). The majority of organic carbon is exported as dissolved organic carbon (DOC); the remainder is particulate organic carbon (POC). Organic nitrogen is also exported primarily in the dissolved form and at the global level riverine export of organic N surpasses that of dissolved inorganic N (DIN)(Meybeck 1982); dissolved organic nitrogen (DON) in rivers comprises 68% of the total dissolved nitrogen (TDN). In the Plum Island System, DON and DOC comprise 80-90% and 92-96% of TDN and TOC, respectively.
Another major source of organic matter in estuaries is intertidal marshes. In marsh-dominated systems along the east and Gulf coasts of the U.S., marsh inputs of organic matter to adjacent tidal creeks often greatly exceed in situ production (Hopkinson 1988).
Estuaries are not merely conduits transporting organic material from rivers to the sea. Estuarine biota use this organic matter, often creating a region of heterotrophy, where respiration exceeds local primary production (Smith and Mackenzie 1987, Hopkinson and Vallino 1995). Most of this respiration is by bacteria (Hopkinson et al. 1989). When supported by terrestrial or marsh organic matter with high C/N ratios, estuarine bacteria compete with primary producers for nutrients (Goldman et al. 1987a,b, Vallino et al. 1996, Wheeler and Kirchman 1986, Kirchman et al. 1989, Tupas and Koike 1990, Toolan et al. 1991, Currie and Kalff 1984). If all the terrestrial organic input was decomposed by bacteria with a 40% growth efficiency, they would use twice the global flux of inorganic N from the land. While much of this organic matter is refractory, a great deal is processed in the estuary and we need to determine its effects on estuarine trophic structure and efficiency.
The delivery of organic matter and nutrients has a major effect on estuarine food webs. For example, the numbers and activity of the flagellates and other members of the microbial loop increased over a eutrophication gradient in Narragansett Bay waters (Hobbie and Cole 1984) and in a freshwater mesocosm (Hobbie and Helfrich 1988). Both zooplankton and benthic animals increased in numbers in response to sewage sludge in a MERL experiment (Oviatt et al. 1987). Several studies have correlated fish production with river discharge and the delivery of organic matter to estuaries (e.g., Sutcliffe 1972, Armstrong 1982). Fish production is generally low (per unit C input) when organic matter loading is high and nutrient availability is low, presumably because of food web inefficiencies (Quiros and Baigun 1985, Welcomme 1985). Blackwater rivers, rich in DOM (usually humics) but low in nutrients, typically have low fish production (Welcomme 1985). Information on the processes underlying these correlations is largely lacking.
The few studies discussed above deal solely with organic carbon; the dearth of data is even greater for organic nitrogen. For example, even the careful budget of Nixon and Pilson (1984) for nitrogen in Narragansett Bay omits the input of organic nitrogen. Studies of DON use in estuaries are generally limited to uptake measurements of labeled amino acids (e.g., Crawford et al. 1973) but amino acids make up only a small proportion of the bulk DON. Only very recently has there been a realization that a significant fraction of riverine DON is labile (Seitzinger, personal communication) and that it may contribute to the nutrition of phytoplankton (Bronk and Glibert 1993). There are no measures of incorporation of DON from land into higher trophic levels of the estuarine food web.
Justification - Hypothesis 2. Variability in Land, Ocean and Atmospheric Forcing: All components of an estuarine ecosystem exhibit variation in space and time in response to external forcings such as changes in climate, river discharge and sea level or in response to internal, biogeochemical and biological processes such as nutrient turnover, population cycles and fish migrations. By comparing time scales of processes, the relative importance of various physical or biological factors in controlling variation in process rates can be assessed and limits placed on the ability of one process to affect another (Hatcher et al. 1987). Processes that occur at similar scales are those likely to interact. Our proposed research on the effect of changes in organic matter, nutrient and water fluxes on estuarine trophic structure and production requires that we evaluate variability in land and ocean forcings and assess the spatial and temporal scales over which the effects of these changes are likely to operate. Understanding the response of these systems to long-term changes in climate, sea level and land cover first requires that we understand and can distinguish short-term variation from long-term trends.
The important external forcings for the Plum Island Sound land margin ecosystem have different scales of variability. Precipitation averages 1.1 m yr-1 but ranges between 0.6 - 1.8 m yr-1. Day to day variability is considerably greater; during a major storm, rainfall can amount to 1/5 the annual mean. River runoff is more variable than rainfall. Daily discharge varies by a factor > 104 over the year and average annual discharge varies 5 fold. Material loading does not vary directly in proportion to discharge; DOC varies to a lesser degree (e.g., 1993-96 annual discharge varied 25% while DOC export varied 20%) while others vary more (e.g., 1993-96 POC export varied 220%). Tidal amplitude averages 2.9 m, but ranges from 2.6 -3.6 m during the neap-spring cycle with storm surges reaching 4.6 m. Sea level has been increasing 2.4 mm yr-1. Mean monthly sea level varies 15 cm over the year but has been as large as 30 cm. Mean annual sea level varies about 13 cm. Other forcings with considerable variability include insolation and temperature.
Estuarine water residence time is a metric that reflects much of the variability in external forcings. Average residence time of water originating in the upper Plum Island estuary decreases from 34 - 5 d as river flow increases from 0.01 to 10 m3 s-1 (Vallino and Hopkinson 1997). In the lower estuary these changes in flow have less effect, with residence time decreasing from 1.2 - 0.6 d. The influence of organic matter and nutrient inputs on the estuarine food web will be dictated by the time scales of important processes relative to water residence time. Examples of these processes include bacterial growth, organic matter decay, particulate organic matter settling, nutrient uptake and phytoplankton growth, and consumer growth relative. Benthic food webs will be less influenced directly by variations in residence time than pelagic webs but nevertheless will be indirectly influenced by changes in salinity and pelagic resources.
We propose to investigate the cause and effect relationships among the physical and biological processes that define estuarine structure and production. We must understand the basic interactions between physical and ecological processes before we can reliably predict the responses of estuaries to changes brought about by human activities (Geyer et al. 1997).
Synthesis - A Trophic Flow Model
We have developed a trophic flow model that synthesizes
our understanding of the effects of organic matter and nutrient inputs
on trophic structure and function. A simplified diagram of the model illustrates
how the major research questions in this proposal fit together (Fig
7).
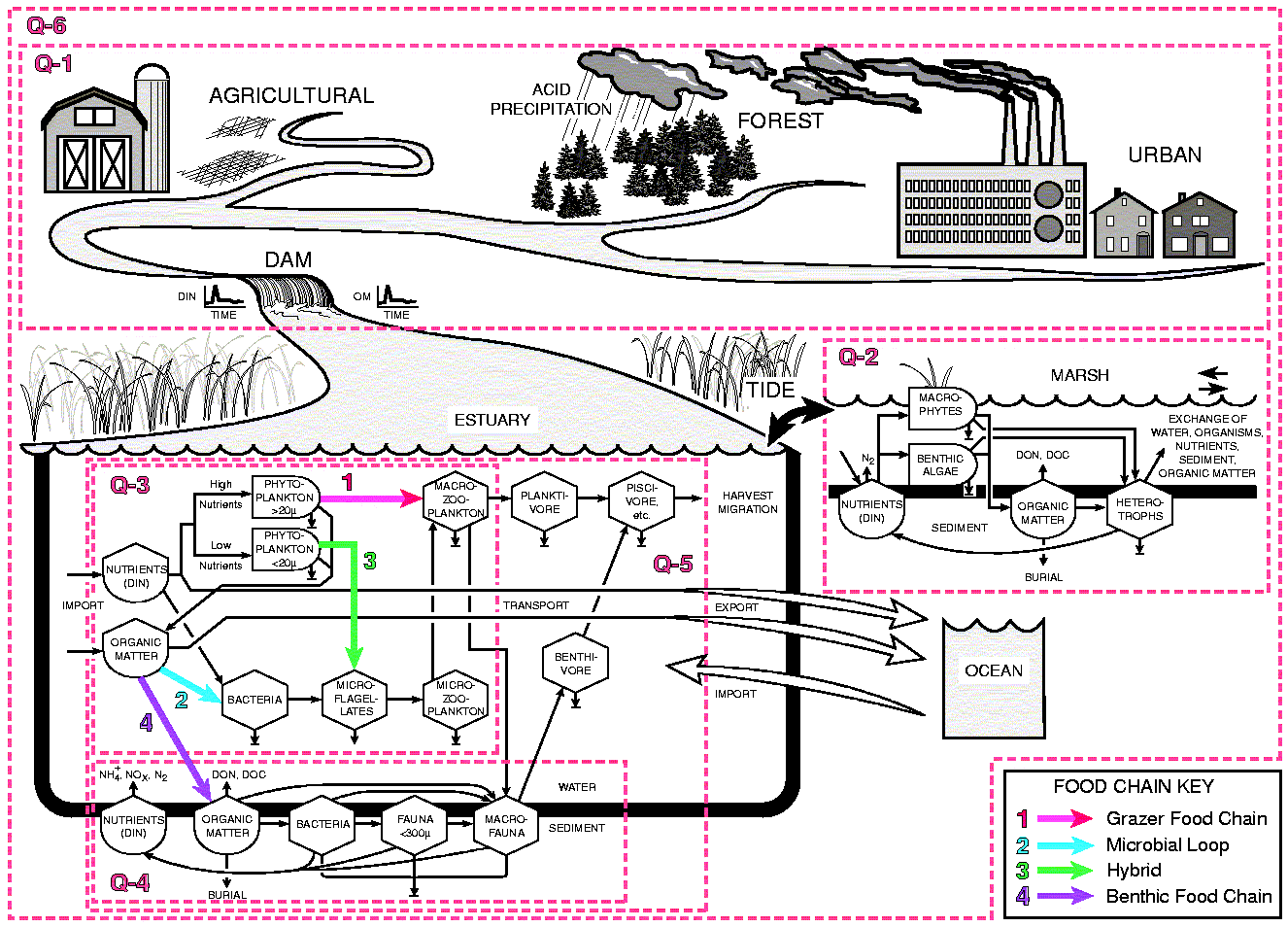
The model illustrates the possible interactions of inorganic nutrients with the quantity and quality of organic matter as it affects trophic structure and trophic efficiency. Four major organic matter and nutrient pathways are portrayed: 1) the classical grazing food chain: inorganic nutrients - phytoplankton > 20 µm - macrozooplankton - planktivores - piscivores, 2) the microbial loop: organic matter - bacteria - microflagellates - microzooplankton - macrozooplankton - planktivores - piscivores, 3) a hybrid of the grazing food chain and the microbial loop: inorganic nutrients - phytoplankton < 20 µm - microflagellates - microzooplankton (the microbial loop) - macrozooplankton - planktivores - piscivores, and 4) a benthic food chain: organic matter - to sediments - bacteria - benthic meio - and macrofauna - benthivores - piscivores.The degree and rate at which organic matter flows through one pathway or another depends largely on the quality of the organic matter being decomposed and the availability of inorganic nutrients. High organic matter loading stimulates the microbial loop at the expense of phytoplankton while high nutrient loading stimulates the grazing food chain. We expect trophic efficiency to be partially controlled by organic matter quality and availability of inorganic nutrients for bacteria.
We used our flow model to evaluate the effects of variations in organic matter and inorganic nutrient loading on trophic structure and production of higher trophic levels including piscivorous fish (Deegan et al. 1994). The analysis test the integrity of our understanding and help us guide research efforts. Two scenarios illustrate the effects of extremes in organic matter and nutrient loading:
Scenario 1. High Loading of Low Quality Organic Matter - Low DIN Loading: the major flow of carbon is through an inefficient microbial loop. Overall trophic efficiency is low; piscivore production is supported mostly by the benthos. The effect of the benthos is to increase the efficiency at which low quality organic matter is processed and transferred through the food web.
Scenario 2. Low Loading of High Quality Organic Matter - High DIN Loading: the major flow of carbon is through an efficient grazing food chain and secondarily through the benthos. Overall trophic efficiency is approximately twice that of scenario 1; piscivore production is supported by grazing and benthic chains. However, the benthos, by shunting more N out of the system via denitrification, causes a reduction in piscivore production.
Proposed Research - We focus our research around five primary research questions. The scope of each question and the manner in which each fits in the overall program is illustrated with numbered boxes around portions of our conceptual model (Fig 7).
Q2. How are tidal marsh processes and their connections to estuarine waters regulated by sea level, storms and water and material inputs from land and sea?
Q3. How does planktonic community structure and production respond to changes in organic matter, nutrients and water fluxes?
Q4. How does benthic use and recycling of nutrients and organic matter vary with changes in water fluxes and the quality and quantity of organic matter inputs?
Q5. How does the structure and function of higher trophic levels
respond to variations in organic matter, nutrients and water fluxes?
The study will be conducted in three land-margin ecosystems. Primary focus is the LMER Plum Island Sound Ecosystem where research will be conducted in watersheds and estuary. Comparative research will be conducted in two other estuaries enabling us to test responses within and across biogeographic provinces and climatic regions. Wells estuary in Maine, and North Inlet estuary in South Carolina, are part of the NOAA National Estuarine Research Reserve network. While all three systems have extensive bordering intertidal marshes and approximately equivalent ratios of marsh to water, the freshwater runoff ranges from being almost insignificant in North Inlet, to moderate in Plum Island and large in Wells.
Biogeographical Perspective: Marine biologists have long realized
the presence of distinct regional distributions of the coastal flora and
fauna. On the basis of diversity and general faunal and floral distributions,
9 geographic provinces have been described for coastal waters of North
America (Fig 8). The provinces are generally related to the 4 climatic
regions of the Atlantic and Pacific Oceans: arctic, cold-temperate, warm-temperate
and tropical (Hayden and Dolan 1976). Past and present coastal LTER/LMERs
are situated in 4 of these 9 provinces (Fig 8)
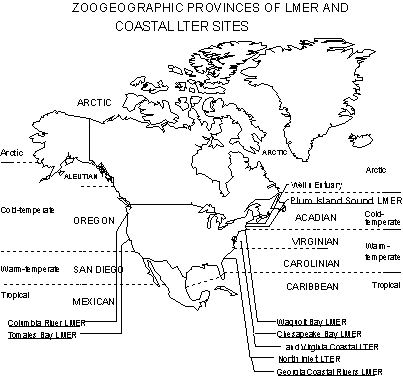
The Plum Island Sound Land Margin System is located in the cold-temperate Acadian Province and would represent a unique addition to the LTER network. Cape Cod represents a very significant boundary between waters to the south which experience extreme seasonal temperature fluctuations (Virginian Province) and waters to the north which are cold year round (Acadian Province). In general the species diversity is low in the Acadian Province. For example, we have found 18 and 28 fish species in Wells and Plum Island estuaries compared to >52 in Waquoit Bay (Virginian Province) (Ayvazian et al. 1992, Buchsbaum 1996). In contrast to the Virginian and Carolinean Province where endemism is extremely low (1% for fish, <10% for mollusks), endemism is high in the Acadian Province (25% for fishes and 30-40% for mollusks)(Briggs 1974). A number of molluscan and algal species found in the Acadian region are arctic or boreal. Fish families common in the Virginian Province and further south such as the Sciaenids (spot, croaker, seatrouts, drum) are replaced by a boreal family, the Gadiformes - hake, cod and haddock, in the Acadian Province. Bay anchovies which are common south of Cape Cod are absent to the North.
The Plum Island System lies at the interface of a thinly soiled,
formerly glaciated New England land mass and the highly productive Gulf
of Maine (Carlozzi et al. 1975). Three watersheds comprise the estuarine
drainage basin: Parker (155 km2), Rowley (26 km2)
and Ipswich (404 km2). The Ipswich River watershed is highly
urbanized with Boston "bedroom" communities encroaching in the headwater
region and the Parker is largely forested. Additional biogeochemical and
ecological background for the system is presented in the Results of Prior
Research section.
Site Integrity: While the watersheds of the Plum Island Sound system are experiencing rapid change in population, economic activity and land use, wetlands both in the watershed and the estuary are well protected by state and federal regulations. Approximately 25% of the Ipswich River watershed has been set aside as conservation land. In addition, most of the estuarine ecosystem is included in the Parker River National Wildlife Refuge. U. S. Fish and Wildlife is enthusiastic about the use of their site as an LTER and has promised full cooperation. Thus the integrity of the system is high and will serve as a valuable laboratory well into the next millennium.
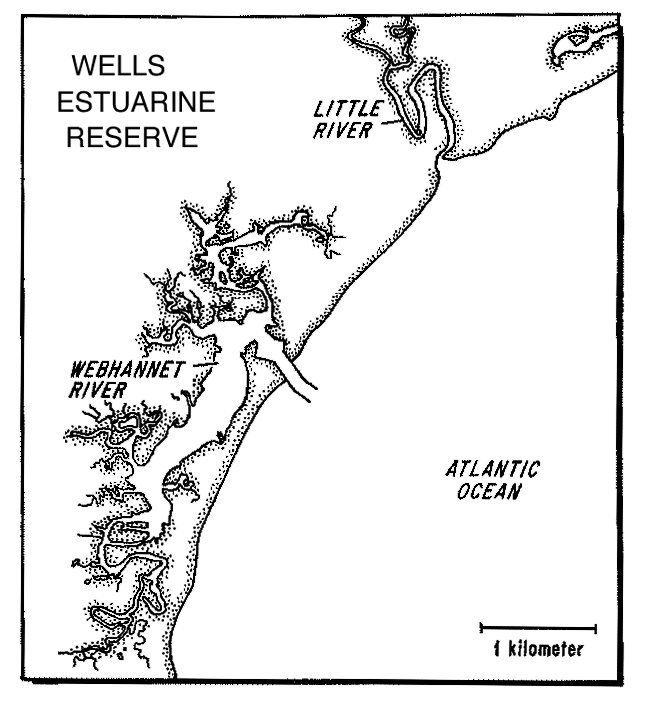
The Wells estuary in southern Maine (Fig 9) is a macrotidal saltmarsh
estuary within the Acadian Biogeographic Province. The estuary is part
of NOAAs National Estuarine Research Reserve network and is in the Rachel
Carson National Wildlife Refuge of the USFWS. The 6.6 km2 estuary
contains extensive salt and brackish marshes with a 4:1 ratio of marsh
to water area. Watershed discharge to the Little River is high relative
to its size thus providing a contrast to the Plum Island and North Inlet
systems. Salinity normally ranges from 5 to 35 psu along the length of
the estuary. Salt marshes are believed to be losing elevation relative
to sea level rise (Kelley et al. 1993) and flood with greater frequency
than Plum Island marshes. Tides are semi-diurnal and range from 2.6 to
3.0 m. The Wells Researve shares many of the same species with Plum Island
(Ayvazian et al. 1992, Deegan et al, in prep.). The Wells Reserve has an
active research and monitoring program focusing on 1) the relationship
between upland land use and estuarine health, 2) the relation between sea
level rise and marsh accretion or degradation and 3) the role of marsh
dominated estuaries in supporting migratory and resident fishes. Co-PI
Dionne will work with the MBL information manager to bring the Reserve
database into compliance with LTER guidelines.
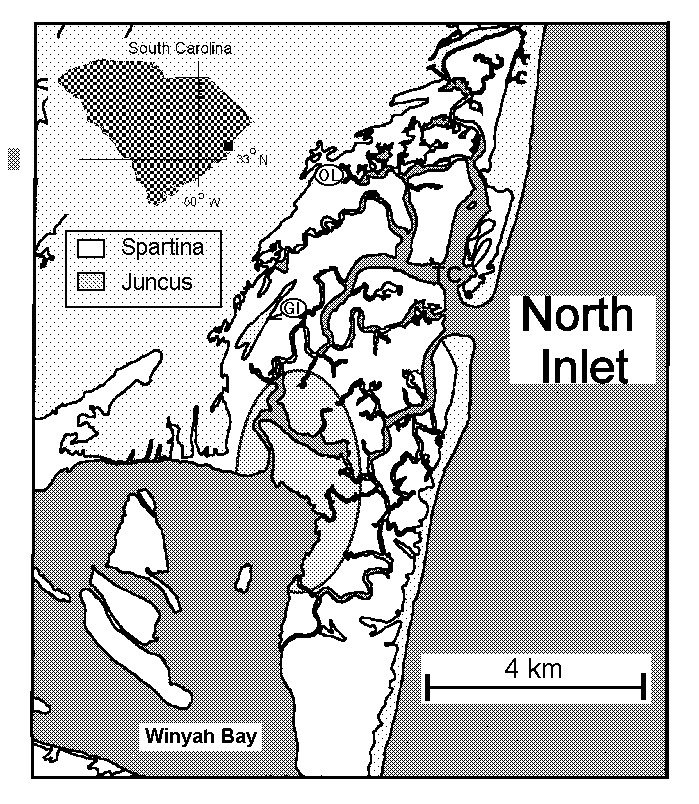
North Inlet, SC (Fig 10) is a tidal saltmarsh estuary in the Carolinian
biogeographical province and is a NOAA National Estuarine Research Reserve.
This site provides a contrast to the macrotidal Parker River and Wells
estuaries in the Acadian province in that terrestrial inputs of water and
materials are extremely low. The North Inlet drainage basin is ~75 km2
but
provides minimal freshwater input. The salinity gradient within the estuary
is only 2.1 psu. The watershed is almost exclusively forested. Estuarine
area is 32 km2 of which approximately 80% is salt marsh dominated
by Spartina alterniflora and 20% is open water. Unlike
the New England systems, marshes do not accumulate peat. The mean tidal
range is 1.7 m. Because surface and ground water inputs of fresh water
are small in comparison to tidal exchanges, the chemistry of the tidal
water within the estuary is dominated by biogeochemical processes occurring
within the creeks and intertidal marshes, and by the magnitudes of material
fluxes through the mouth. Summertime algal blooms occur in response to
high rates of nutrient remineralization (Lewitus et al. submitted) as opposed
to the situation in the Plum Island system where summer blooms are due
to riverine nutrient inputs. North Inlet was an LTER site from 1980-1993.
The extensive LTER database, which is continued by NOAA and an LTREB grant
to Morris, includes data ranging from climatology to higher trophic levels.
Differences in climate, biogeographic provinces and land runoff make this
an excellent comparative system to the Wells and Plum Island systems.
PROPOSED RESEARCH
QUESTION 1. What is the magnitude and temporal pattern of organic carbon and nitrogen and inorganic N loading from watersheds to the estuary? (Q1 in Conceptual Model, pg 2-4)
RATIONALE - The temporal pattern of organic matter and nutrient loading
to estuaries is primarily driven by the daily and seasonal variations in
discharge. In the Acadian Province precipitation is relatively evenly distributed
over the year, however due to snowmelt and summer evapotranspiration, discharge
normally peaks in late winter to spring with relatively low discharge during
the June to October period (Sammels 1967) (Fig 11). However, this low flow
season can be punctuated by runoff from tropical storms which can yield
as much discharge in a day as normally occurs in a month or more. Both
the daily and annual temporal patterns are significant for the estuarine
ecosystem because the fate of organic matter and nutrient inputs depends
critically upon the state of the biological system (warm growing season
versus cold season) and the residence time of watershed-derived material
in the estuary once it arrives (Hopkinson and Vallino 1995). When discharge
is high (residence time is short) and temperatures are low, most dissolved
organic matter will pass unused through the estuary to the ocean. However
bioassays indicate up to 20% utilization if residence time is weeks and
temperature is moderate (Uhlenhopp et al. 1994, Hopkinson et al. submitted).
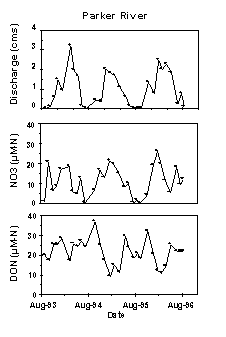
Superimposed on the variability in discharge is a seasonal cycle
of concentration for DOM, POM and nutrients. For rivers entering Plum Island
Sound, the general pattern is for PO43-, NO3-,
and NH4+ to be higher during winter and lower during
the summer growing season. Phosphate levels are normally low, seldom exceeding
1 mM. Ammonium rarely exceeds
2 mM whereas NO3- levels
vary from a 1-2 µM in summer to 10-30 mM
in winter (Fig 11). Much of this variability in nutrient concentration
is undoubtedly linked to biotic activity in the drainage with NO3-
levels increasing when biological activity is low (Likens et al. 1977).
The DON concentrations are somewhat less variable over time ranging from
about 10 to 40 mM (Fig 11). The lability
of DOM to microbial use depends on land use, with urban and agricultural
catchments yielding more labile DOM with lower C:N ratios than forested
catchments (Uhlenhopp et al. 1994). Loading to the estuary displays a strong
seasonal pattern mainly driven by discharge but amplified by the strong
seasonality in NO3- concentration.
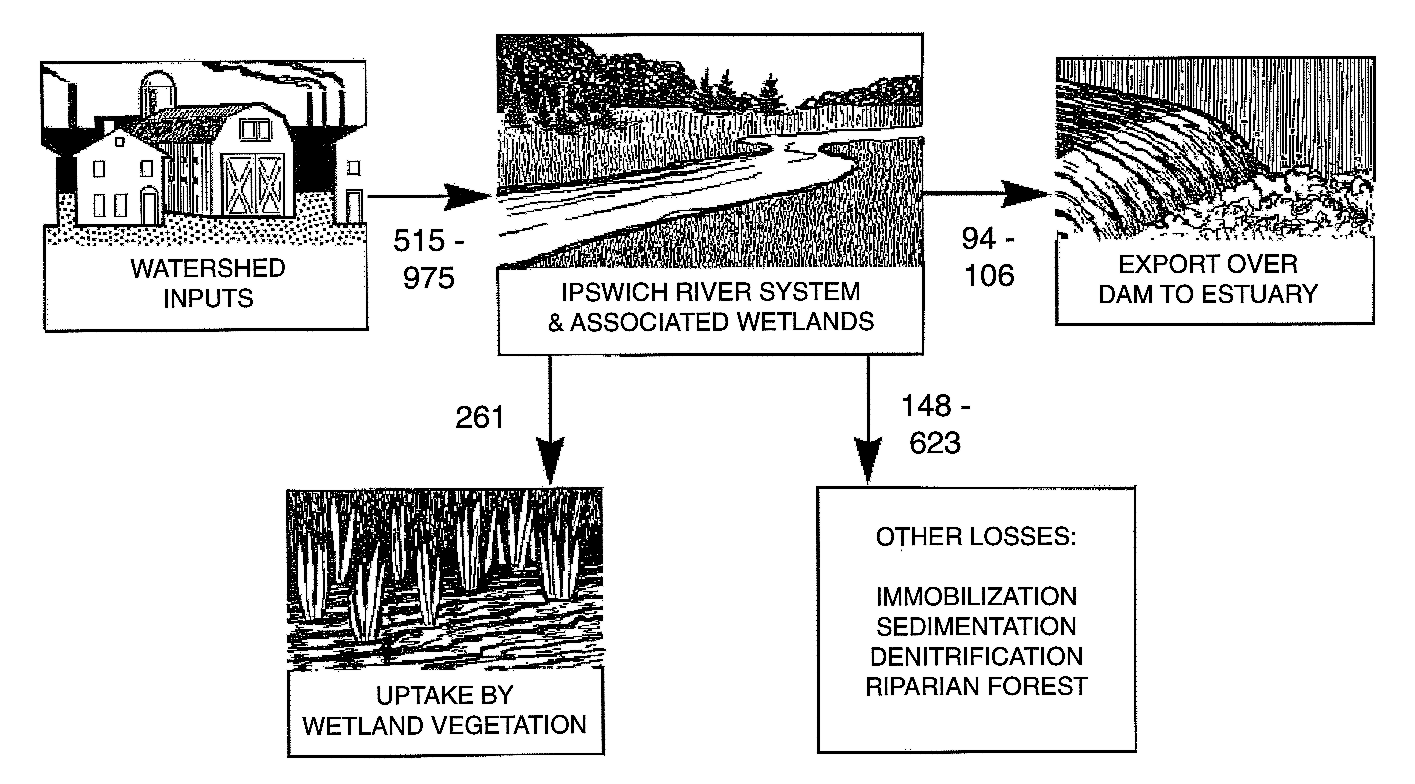
We have begun watershed modeling using our data on the relationships between land use and export of organic matter and nutrients to rivers in order to develop nutrient budgets for subcatchments and the major watersheds. Landuse maps were constructed from digital town maps (MassGIS 1992) for the years 1971 and 1985 (1991 in production) (Fig 1, pg. 1-1). Hydrologic and nutrient loading related parameters were determined for each landuse type in the watershed. Urban subcatchments had the highest NO3- concentrations whereas farm subcatchments had higher NH4+ and DON concentrations than forested catchments (Fig 2, pg. 1-1). Subcatchment results were scaled to the watershed for annual periods with hydrologic models (TR-55 (SCS 1986), Brook6 (Federer and Lash 1978, Miller 1988) and GWLF (Haith et al. 1992). The results of this scaling were disconcerting because loading from small catchments to the rivers was calculated to be 515-975 Mg y-1 but only about 100 Mg y-1 was measured as export to the estuary (Fig 12). This finding highlights a significant feature of the nitrogen cycle of these watersheds. There is major retention of N in the river network including riparian vegetation, fresh marshes, swamps, and ponds, which we must account for in order to extrapolate from small catchments to estuarine loading from rivers.
Specific Research Questions: 1a) What is the temporal pattern of nutrient and dissolved and particulate organic matter output from catchments with different land covers?
The results of small catchment monitoring indicate significant differences in nutrient concentrations, in organic matter concentrations, and in organic matter lability that correlate with land cover and use (Fig 2, pg. 1-1, Uhlenhopp et al. 1994, Finn and Hopkinson 1997). Our preliminary efforts require testing and confirmation at additional sites with higher intensity of sampling and monitoring. For example, storm discharge and chemistry were monitored on selected storms but not routinely and sampling dates were widely spaced.
Approach: We will continuously monitor discharge and collect weekly plus intensive storm water samples for nutrients and organic C and N with ISCO automated samplers on representative subcatchments dominated by urban, farm and forest landuse. Parallel and simultaneous monitoring and water chemistry measurements will be made at the dams where the Ipswich and Parker Rivers discharge to the estuary. Samples will be assayed for pH, alkalinity, NO3-, NH4+, PO4º, DOC, DON, POC, PON, TP and TSS. These data will be used to calibrate the Brook and GWLF hydrology and nutrient loading models for subcatchments. Periodic extensive sampling of 6 subcatchments in each land use category will be used to validate model predictions and to test if the intensively sampled catchments are representative of their landuse class. This sampling will continue for the 6 years of the grant to assess both within and across year variability in export from each land use class and from the major Ipswich and Parker watersheds. Modeling - This information on the subcatchments with different land use will be used to calibrate the GWLF to calculate loading to the mainstem river.
1b) To what extent does riparian and in-stream retention and processing affect the timing, magnitude and composition of organic matter and nutrient delivery to estuaries?
If river corridor processes were not as effective in retaining nutrients, nitrogen loading to Plum Island Sound would be 7 times higher (Fig 12), an increase likely to have significant impact on the N-limited estuary. Processes potentially responsible for N retention and loss include uptake by swamp and fresh marsh vegetation, algal uptake, sedimentation and denitrification (Seitzinger, 1988, Billen et al. 1991, Howarth et al. 1996). We must assess the location and magnitude of the riparian and riverine processes responsible for retaining N to find out if these are permanent or temporary N storages and sinks or if they are nearing saturation.
Approach: In order to locate river reaches most active in removing nitrogen we will perform intensive longitudinal surveys of nitrogen concentrations in the Ipswich and Parker Rivers seasonally. Sampling stations will be located to test the impact of particular habitats on net N retention and mineralization. At each site, we will measure discharge, pH, NO3-, NH4+, PO4º, DON, DOC, PON and POC. For each reach we will perform short term solute addition experiments with both a conservative tracer and NO3- enrichments to estimate seepage, nitrate uptake rate and uptake length. In several reaches and fringing habitats that appear active in N uptake, we will set up feldspar marker horizons and sedimentation plates to estimate long-term sediment and organic matter accumulation. Once the reach most active in N retention is located, we will perform an intensive study of the pathways and rates of N cycling in that reach. A one-month tracer addition of 15N-labelled NO3- will facilitate the assessment of the N flow pathways and rates of turnover of stocks of N in the river ecosystem (Peterson et al., in press). Our experiments will follow the established protocols used for the NSF-funded Lotic Intersite Nitrogen Experiments (LINX, Webster, Meyer, Mulholland, Peterson, PIs) including conservative tracer additions and modeling (Bencala and Walters 1983, Peterson et al. in press). The design of this tracer experiment will be adjusted to optimize the accuracy of the N flux estimation according to the guidelines recently developed by Wagner and Harvey (in press).
15N-labelled NO3- will be added continuously for one month via a peristaltic pump at a rate sufficient to achieve a 500 per mil enrichment of the NO3- in the river. Discharge, DO, conductivity and temperature will be monitored continuously with YSI probes and data logger. Transects of samples downstream from the tracer addition site will be collected on days 1, 3, 7, 14, 21, 28 during the addition and at days 1, 7, 14 and 28 after the additions. Samples will include NH4+, NO3-, N2, DON, PON, benthic FPON and CPON, algae, marsh plants, and consumers. The samples will be analyzed for N content and tracer 15N content. Modeling - The stream nitrogen cycling model currently used on the LINX project will be employed to analyze the isotopic field data for the intensive study reach . We will also apply the EPA Water Quality and Analysis Simulation Program (WASP5) to the entire main river channel. This model represents the river as a network of nodal points connected by channels that route water and materials. Coupled with our existing generalized river metabolism model, the nodal model will enable us to route, process and deliver materials to the estuary.
QUESTION 2. How are tidal marsh processes and their connections to estuarine waters regulated by sea level, storms and water and material inputs from land and sea? (Q2 in Conceptual Model, pg 2-4)
RATIONALE - Estuarine marshes act as filters adjacent
to tidal creeks and bays that intercept, transform and modify material
inputs from land and ocean. Marshes are also important sinks for atmospheric
CO2 and sources of organic carbon to coastal waters and food
webs. As well as serving as net sinks for inorganic nutrients, marshes
are leaky of nutrient inputs to some extent and thereby can serve as important
sources of inorganic nutrients to adjacent tidal waters. Lastly marshes
provide habitat critical to the development of many species of fish and
shellfish, many of which are commercially valuable.
Sea level, tides, storms and material inputs regulate the intensity of these marsh processes and the connectivity between marshes and adjacent tidal waters. Storms and tidal currents provide the energy to resuspend, erode and transport materials within watersheds and the coastal zone. Sea level determines connectivity, especially the frequency and duration of marsh flooding. Flood tides are essential for water borne materials to be imported to marshes. Marsh sediment accretion is a dynamic process (Krone 1985, Reed 1989, Cahoon 1994, Cahoon and Reed 1995, Cahoon et al. 1995) of extreme importance to the coastal zone, as it determines the ability of intertidal marsh areas to keep pace with rising sea level. Absolute changes in surface elevation are determined by sedimentation, seasonal cycles of belowground production and decomposition, sediment dewatering, and compaction. There are complex feedback mechanisms among these processes, such as feedback between marsh primary production, sea level, tidal flooding, and porewater salinity (Morris 1995), which in turn influences sedimentation due to changes in the density of vegetation (Leonard and Luther 1995).
Tides, sea level and freshwater have significant effects
on primary production (Morris and Haskin 1990), plant community composition
(Olff et al. 1988), sediment salinity (Morris 1995), and habitat availability
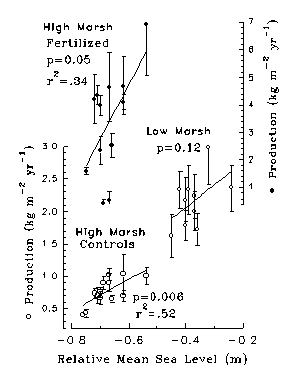
(Rozas 1995). Salt marsh primary production in North Inlet, SC is positively
correlated with interannual changes in mean sea level (Fig 13).
At low mean sea level, flood frequency is reduced, sediment salinity rises
with evaporative water losses, and productivity of Spartina declines.
Changes in mean sea level during mid-summer vary by as much as 20 cm from
one year to the next due to variations in the thermal expansion of the
ocean, atmospheric pressure, and prevailing winds.
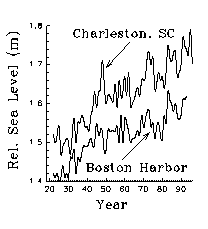
Interannual changes in mean sea level are at least regional
in scale. Long-term variations in mean monthly sea level from Atlantic
and Gulf Coast NOAA tide gauges are highly correlated (Morris et al.1990).
The correlation between mean annual sea level in Charleston and Boston
is 0.82 (Fig 14). It is not known if estuarine primary production
responds regionally to changes in mean sea level, but these correlations
suggest that it could. The entire coast may have an ecological rhythm that
varies with mean sea level.
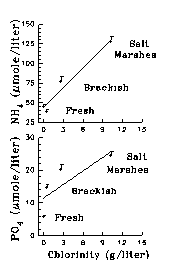
The dynamic balance between tides, sea level and river runoff affects salinity distribution in an estuary which in turns influences numerous biogeochemical processes. As the salinity varies along the axis of an estuary, the availability of PO43- and NH4+ varies as well. For instance, at the fresh water end of an estuary, DOC sequesters soluble reactive phosphorus (SRP) and reduces its bioavailability (Sholkovitz 1976, Jones et al. 1993, Fox et al. 1986, Froelich 1988). Approaching the sea, the contribution of Fe-P to total P diminishes while that of Ca-bound and easily exchangeable phosphate increases (de Jonge and Villerius 1989, Lebo 1991, Zwolsman 1994). Consequently, the concentration of free PO43- increases with salinity in marsh sediments (Fig 15).
Salinity also has a major effect on the sorption and release of NH4+ from sediments. Generally, the ratio of exchangeable: dissolved NH4+ will increase as salinity decreases (Mackin and Aller 1984, Rosenfeld 1981, Seitzinger et al. 1991). Exposing freshwater sediments to sea water, which could happen during a period of low flow for example, results in a significant release of NH4+ (Gardner et al. 1991). These processes appear to elevate the concentration of free NH4+ in salt marsh pore water relative to fresh and brackish marsh sediments (Fig 15), and may signify a fundamental difference in the nutrient source and sink functions of fresh and salt marshes.
Sea level also influences the drainage of marsh porewaters by directly affecting the hydraulic gradient between tidal creeks and the marsh water table. During times of low mean sea level, there is increased drainage of nutrient-rich water from marshes due to a steeper hydraulic gradient. There is evidence for this effect In North Inlet, SC where there are significant negative correlations between monthly sea level anomalies and nutrient concentrations or with chlorophyll.
Specific Research Questions: 2a) How are sediment accretion and primary production in tidal marshes altered by changes in freshwater discharge, material inputs and sea level?
Interannual changes in mean sea are at least regional in scale (Fig 14), but it is not known if estuarine primary production responds regionally to changes in mean sea level, and it is not known how microalgae and organic matter accumulation respond. In North Inlet, Morris has demonstrated enhanced organic matter decomposition that overrides the enhancement of primary production causing a net loss of organic matter. Rybczyk et al (1996) found little indication of enhanced rates of organic matter decomposition or accumulation when studies were conducted over a long enough interval (>1 or 2 yrs).
Approach: We will monitor physical variables, porewater nutrients, plant and soil stores of N and P, sedimentation, accretion and primary production of macrophytes and microalgae in control and N&P fertilized sites along a salinity gradient in the Plum Island Sound estuary and at Wells, ME, and North Inlet, SC. A description of long-term fertilization experiments is presented later in the proposal under the heading "Research Approach - Long Term Experiments" (pg. 2-20). Net aboveground primary production of macrophytes will be monitored using a non-destructive census technique (Morris and Haskin 1990). Marsh sediment elevation will be monitored from permanently installed SET platforms (Boumans and Day 1994) that are presently used at North Inlet and Wells and feldspar marker horizons (Cahoon and Turner 1989). Sedimentation will be measured with sediment collectors: (Reed 1989, Boumans and Day 1994) 10 replicate ceramic plates placed near SET platforms.
2b) How do indicators of organic matter and inorganic nutrient exchange between marshes and estuarine waters respond to variations in freshwater discharge and sea level?
The transfer of organic matter and inorganic nutrients to adjacent tidal creeks represents an important source of energy, C and N to pelagic and benthic food webs. The Plum Island Sound estuary is markedly heterotrophic, beyond that sustainable by riverine inputs of OM. Thus theres little doubt that marsh-derived organic matter is exported and metabolized to some extent (Peterson et al. 1994).
Approach: Direct quantification of marsh organic matter export has been attempted several times (Wiegert and Chalmers, North inlet flumes). Results unfortunately are equivocal and direct quantification requires herculean efforts. We will monitor "indicators" of export. On the marsh we will monitor hydraulic head between marsh and tidal creeks, measure hydraulic conductivity, and monitor porewater nutrients. Measurements of depth profiles of pore water nutrients in marshes provide key evidence about temporal and spatial patterns of the source-sink function of marshes. These data will be paired with tidal, salinity, discharge, rainfall, river water chemistry, and productivity data. These indicators have not previously been examined together within the context of an ecosystem study. Depth profiles (to 1 m) of NH4, SRP, sulfide, and major cations and anions will be monitored in marshes along the Parker R., Wells, and North Inlet using diffusion samplers placed in the sediment and left to equilibrate for a period of one month.
QUESTION 3. How does planktonic community structure and production respond to changes in organic matter, nutrients, and water fluxes? (Q3 in Conceptual Model, pg 2-4)
RATIONALE - We will determine the amount of watershed-derived organic matter entering the estuary (Q1) as well as indicators of marsh-derived organic matter (Q2). The next question, the focus here, is on the use of this material by organisms and the effect of changes in the inputs.
Bacteria, the key element in the use of organic matter
in the estuary, are able to utilize dissolved and particulate organic matter
(DOM and POM) from upland watersheds, from algal blooms in the upper estuary,
from salt marshes, and from the ocean (Peterson et al. 1985). The DOM and
POM from these sources are difficult to distinguish chemically but more
important, only a fraction of the material may be decomposed and used by
the bacteria. An efficient way to investigate the organic matter actually
used is through stable isotopes, a technique pioneered for estuarine research
by Bruce Peterson and Brian Fry at the MBL. Based on data from the Parker
River (Fry et al. 1992, Coffin et al. 1989, Fry et al. 1993, Peterson et
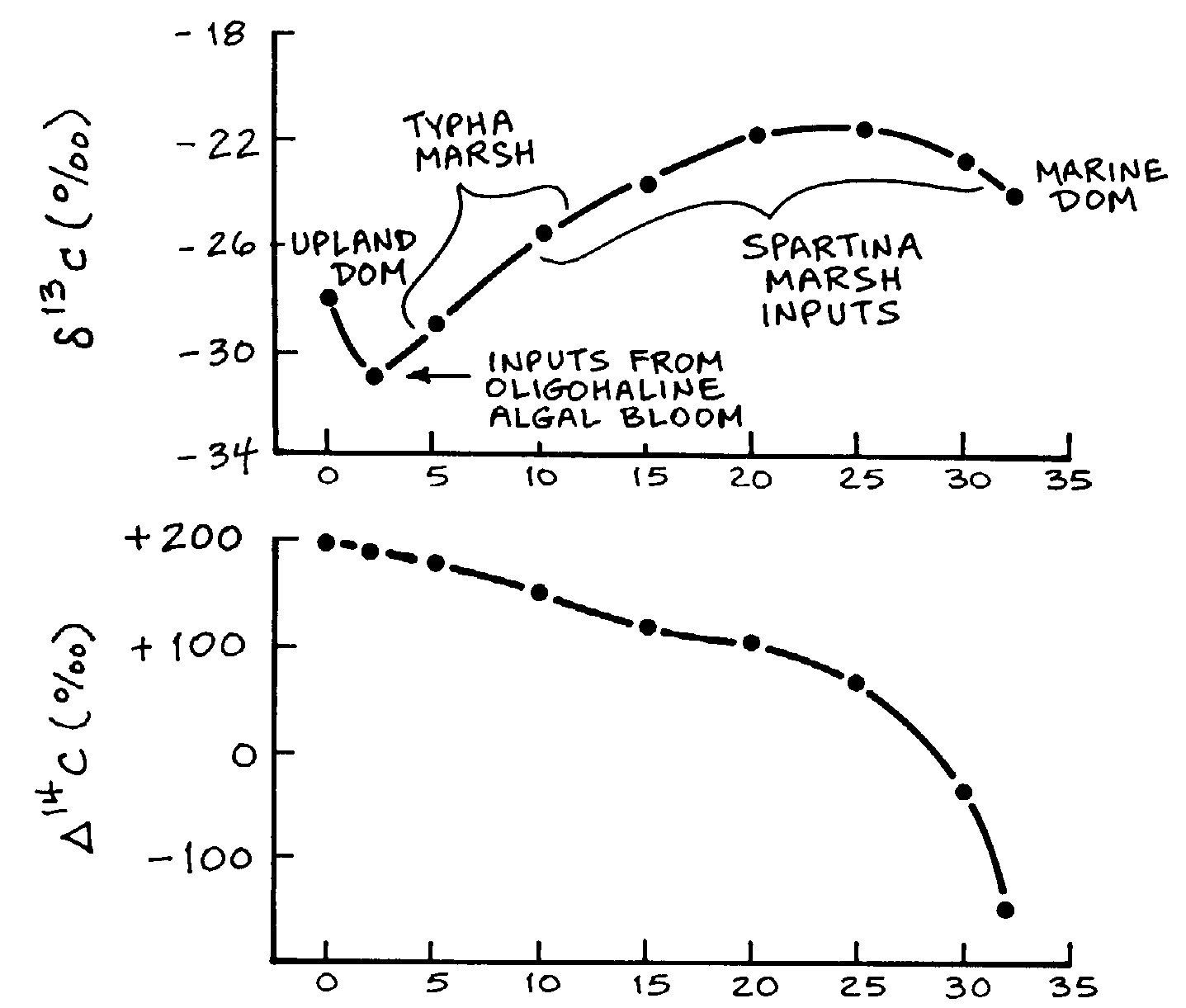
al. 1994, Hullar et al. 1996), a conceptual diagram of the d13C
of the DOM shows changes as DOM from different sources enters the river,
is transformed and transported downriver, and exchanges with the marine
DOM (Fig 16). The endmembers of DOM from the upland and DOM from
the ocean have d13C values of -28
and -22, respectively. DOM from an oligohaline algal bloom has a d13C
value of -44, from a freshwater Typha marsh has a d13C
of -26, and that from a Spartina marsh has a value of -13. If there
were only two sources, the upland DOM and the marine DOM, then the conservative
mixing relationship would be an upward curving line running from -28 to
-22. On the diagram, the d13C of
the DOM does not follow this path at all but, instead, the bulk DOM reflects
each of the inputs that enters the estuary as it mixes with the bulk DOM
from upstream and is transformed. Although we have not yet exploited the
d14C
signal, we predict a strong gradient of this signal along the estuary (Fig
16).
Peterson (in prep.) has developed an inverse modeling
approach (similar to Vezina 1989) to estimate the inputs and exports of
DOM from various sources including marshes, phytoplankton, and ocean. The
model is based on the measured amount and d13C
of the DOM entering the estuary from the river, of the measured microbial
consumption based on bacterial production, of the measured d13C
of the DOM, and of the d13C of estuarine
bacteria grown in filtered estuarine water (method in Hullar et al. 1996).
For one time period (see Table 1) most of the organic matter (as dissolved
organic carbon or DOC) came from the salt marsh; also, most of the bacterially-active
DOM came from the salt marsh. Despite the size of the input from the uplands,
the amount consumed by bacteria was very small, presumably because the
DOC had already been modified by bacteria in the soil and in the river
before entering the estuary. This approach provides an estimate of the
bacterial use of different sources unobtainable in any other way. The additional
measurement of d14C will further
constrain our model.
| Table 1. Model estimates of DOC inputs, microbial consumption and export to the coastal ocean for the Parker River-Plum Island Sound during July-September (rates averaged over an entire spring-neap tidal sequence). | ||||
|
|
||||
|
|
|
|
|
|
| River | 2.9 | 0.1 | 2.8 | |
| Fresh Marsh | 0.8 | 0.7 | 0.1 | |
| Phytoplankton | 2.3 | 1.8 | 0.5 | |
| Salt Marsh | 18.0 | 5.8 | 12.3 | |
| Ocean | 0.6 | 0.2 | 0.4 | |
Another key interaction in the estuary is the competition for nutrients between bacteria and phytoplankton. When organic matter, dissolved and particulate, is low in N and P, then bacteria will take up inorganic nutrients (Wheeler and Kirchman 1986, Goldman et al. 1987a) and thus compete with algae (Rigler 1956, Rhee 1972). In fact, Thayer (1974) found that microbial immobilization of N and P during decomposition of low quality particulate organic matter limited phytoplankton productivity in North Carolina estuaries via nutrient limitation. In the same way, Hopkinson et al. (1989) and Goldman et al. (1987a) showed that net bacterial N regeneration occurred only when the substrate C/N ratio was below 10-12 to 1. These empirical results agree well with the theoretical model developed on this project by Vallino et al. (1996). Empirical results from the Parker River bioassay incubations of terrestrial DOM with estuarine bacteria (Fig 3, pg. 1-3) revealed a seasonal effect: N was remineralized during low flow periods in the summer when DIN was low in concentration and immobilized during high flow when DIN was high. Under certain circumstances algae are also able to gain nitrogen by enzymatically removing the amino group from DON (Bronk and Glibert 1991).
In our LMER research we carried out a large carboy experiment (Norrman et al. 1995) and a mesocosm experiment (10 m3) looking in part at the competition between algae and bacteria for nutrients. Vallino used the resulting data to validate a model he had constructed based upon the literature. This is itself a big step forward because almost all process-based models of planktonic foodwebs have never been tested. The model could not be validated and our conclusion was that science still lack data on the fundamental relationships among nutrients, organic matter quality including DON, the decomposition process, bacteria, and algae. Accordingly, small-scale carboy-sized experiments need to be carried out to investigate these processes and their controls. The data on relationships will be used in an improved model that will include the effect of water residence time on algal productivity.
To translate the impacts of the changing organic matter quality, nutrients, and water residence time to higher trophic levels of the plankton, the key group to investigate is the zooplankton. Several studies have correlated fish production with river discharge and the delivery of organic matter to estuaries (Sutcliffe 1972, Armstrong 1982) but there is no information on the processes involving zooplankton that underly these correlations. In an attempt to examine the interactions between nutrients and organic matter, our LMER project carried out experiments in which organic matter from land plus nutrients, and nutrients alone, were added to 10 m3 mesocosms in Woods Hole. There was a zooplankton response to added DOC (Fig 17); we found the highest numbers of zooplankton (1600/liter) in the DIN + DOC bag.
Specific Research Questions: 3a) What is the
spatial and temporal variation in the source of dissolved and particulate
organic matter (from land, oceans, macrophytes, and algae) supplied to
and used by pelagic bacteria?
Approach: We will use the different isotopic labels to differentiate the sources by analyzing the d13C in the DOM and POM in one study per year along the Parker River. For each study, two sets of nine samples will be collected over the river; the sets will be 28 days apart so they are at the same point of an entire spring-neap tidal cycle. d13C of bacteria will be analyzed with techniques of Hullar et al. (1996) using natural bacteria grown on filtered natural waters. As a second tracer (Fig 16), we will also analyze the d14C using the Woods Hole (WHOI) Accelerator Mass Spectrometer, directed by John Hayes. Each year we will sample at a different time of year to build up a seasonal picture. Inverse modeling will be used as previous described.
3b) How does the quality and quantity of organic matter mediate competition between bacteria and phytoplankton for nutrients?
Approach: We use experiments to develop mathematical relationships for the various processes involved. Replicate 7-d experiments in 10 liter carboys containing 250 m m-filtered water will be used to test the interacting effects of added nutrients, light, added organic matter of high and low quality (e.g., POM and DOM from different locations in the estuary) on algal and bacterial growth and on availability of the organic matter to decomposition. Daily measurements include NO3, NH4, DOC, DON, numbers and production of bacteria (leucine method), flagellates, and phytoplankton numbers and production (14C). A process-based model will be assembled.
3c) How does production of zooplankton and fish differ according to changes in water residence time, organic matter inputs, and nutrient supply?
Approach: Experiments will be run for ~30 days in mesocosms (4 ea. 10 m3 plastic bags incubated at Woods Hole) in the same way as a previous LMER experiment. The focus will be on the production of zooplankton under various conditions of organic matter inputs and nutrient supply. Treatments will consist of additions of terrestrial litter leachate, inorganic nutrients to stimulate the release of OM from phytoplankton, or both. Measurements will include zooplankton numbers and biomass, stable isotope values of zooplankton, phytoplankton, and added organic matter. Data assimilation techniques will be used to develop and tune the model so it will apply to a range of estuarine environments. Once the dynamics of the mesocosm observations are captured, then the effect of residence time and of implications for fish growth will be investigated with the model.
QUESTION 4. How does benthic use and recycling of nutrients and organic matter vary with changes in water fluxes and the quality and quantity of organic matter inputs? (Q4 in Conceptual Model pg 2-4)
RATIONALE - The benthos is an important site of organic matter mineralization in estuaries, consuming substantial quantities of oxygen and releasing nutrients back to the overlying water (Nixon 1981, Boynton and Kemp 1985). However, it is now well accepted that the benthos also serves as both a short-term and a permanent sink for nutrients through adsorption, denitrification and burial. Because N is a nutrient limiting primary production (Ryther and Dunstan 1971), many studies have focused on the importance of denitrification (Seitzinger 1988, Jorgensen and Sorensen 1988). In an examination of the elemental budgets of nine estuaries Nixon et al. (1996) concluded that 30-65% of the N entering the estuaries was denitrified or buried. In most estuaries P retention appears to be lower, but in some systems, such as Chesapeake Bay, P retention is greater than N retention (Nixon et al. 1996). Because some estuaries exhibit spatial or seasonal shifts in N vs. P limitation (DElia et al. 1986), estuarine studies need to consider processes that control the location, timing and magnitude of benthic N and P fluxes to overlying water.
The importance of the benthos in decomposing organic matter is usually evaluated by the percentage of total ecosystem respiration that occurs in the sediments. This measure may underestimate the role the benthos plays in estuarine productivity and trophic structure. Because of the much longer residence time of particles in the sediment than in the water column and alternating redox conditions (Aller 1994), the benthos plays a key role in the processing of low quality organic matter with high C/N ratios such as terrestrial and marsh detritus. As some organisms are capable of obtaining a portion of their N and C directly from detritus (Cammen 1980) the benthic food web may be shorter and more efficient (Newell 1982, Findley and Tenore 1982); the importance of this pathway will be a function of the quality of the detritus. Our flow model analysis (see pg. 2-4 to 2-5) suggests that the benthos increases the overall efficiency at which low quality organic matter is transferred through the food web to higher trophic levels. However, because of sedimentary denitrification, and the high C/N of terrestrial and marsh detritus being processed, the return of N to the water column from the benthos is low relative to the C and N input. Therefore, another effect of the benthos is to drive the ecosystem towards heterotrophy.
The importance of the benthos to ecosystem productivity and trophic structure also changes as conditions in the overlying water change. Under conditions of very low salinity, less of the N and P mineralized from organic matter reaches the overlying water than under saline conditions. This is because both denitrification (Seitzinger et al. 1991) and phosphorous adsorption (deJonge and Villerius 1989) are greater under low salinity. At the marine end of the estuary, elevated sulfide levels in saline porewater inhibit denitrification, thus increasing the flux of remineralized N (Joye and Hollibaugh 1995). Hence, both the benthos and the marsh (Q2, pg. 2-10) should contribute greater quantities of nutrients to the water column during years of low runoff and/or higher relative sea level.
Specific Research Questions: 4a) How do benthic nutrient fluxes and metabolism in different regions of the estuary respond to variations in the quality and quantity of organic matter inputs?
The sources of organic matter to the estuary change along its length. We will measure how total benthic respiration and inorganic nutrient and DOM fluxes change with variations in the quality and quantity of organic matter inputs along the salinity gradient in the estuary. Both the quality of the organic matter (low or high C/N, vascular vs. planktonic detritus) and the environment in which it is decomposed (electron acceptor availability) will be important factors affecting the dynamics of nutrients and organic matter in sediment.
Approach: Long term measurements - We will measure benthic fluxes twice a year at the upper and mid estuarine sites. We will look for changes in fluxes as watershed inputs vary through natural variations in river discharge (at the upper site) and as marsh inputs vary with variations in sea level (at the mid-estuary site, see Q2, pg. 2-10). Process studies - We will compare benthic fluxes from intact sediment cores from the low and high salinity site where organic matter is added either as terrestrial POM, marsh detritus, or planktonic detritus. Comparisons from two salinity zones will enable us to compare fluxes from sediments where sulfate reduction or methanogenesis is the dominant mode of anoxic metabolism. The organic additions will be grown for the experiment and enriched in 14C to facilitate tracing the carbon into the benthic food web. 3) Mesocosms - We will compare benthic respiration and nutrient fluxes against a control in mesocosms where the water column receives either inorganic nutrients, terrestrial DOM , or DOM+nutrients (Q5 pg. 2-17).
4b) What is the relative importance to higher benthic trophic levels of carbon and nitrogen from organic matter of different qualities?
The flow of carbon and nitrogen may become more uncoupled in benthic food webs as the quality of organic matter decreases. Field surveys of organism C and S stable isotopic signatures indicated preferential utilization of marsh-derived organic matter by benthic organisms. Use of terrestrial detritus was equivocal (Deegan and Garritt 1997). However, in a whole system N-15 addition, it was apparent that some organisms which appear to be tightly linked to marsh detritus for C also exploit phytoplankton for a portion of their N requirements. We do not have any information on year to year variability in food sources or comparative measures of growth on different types of detritus.
Approach: Long term measurements - We will collect samples of the bivalves, Mya and Geukensia, at the mid estuarine site annually, and use variations in their d13C, d15N and d34S values to assess differences in the importance of phytoplanton vs marsh detritus to benthic filter feeders. This gives us another indirect measure of how year to year variability in hydrology affects the relative contribution of marsh detritus and phytoplankton to benthic food webs (Q2 pg. 2-10). Process studies - We will add the isopod, Cyathura polita, to cores receiving different sources of organic detritus as described in 4a. This hardy, benthic detritivore is common in the upper to mid-estuary and has a broad salinity tolerance. The experiments will be carried out in the dark with filtered overlying water to eliminate water column sources of nitrogen. Analysis of survival, growth, and the 14C content of the animals measured at the end of the experiment will indicate efficiency of utilization of the various organic matter sources.
4c) How do variations in the salinity distribution in the estuary determine the magnitude and timing of nutrient release to the water column?
During 1993 and 1994, we found that mid- to late summer inorganic nitrogen fluxes from sediments in the upper estuary were higher than could be supported by benthic remineralization. This suggested that during this time of the year denitrification was of minor importance and that some process in addition to immediate decomposition was contributing to the large N release. This phenomena did not occur in 1996, a year of high runoff and low estuarine salinity, ie., < 6 psu. We now hypothesize that as porewater salinity increases during summer, exchangeable ammonium is released from sediment particles causing high ammonium fluxes. A single measurement of adsorbed ammonium in sediments in spring showed sufficient ammonium on particles that if released could account for the high summer fluxes we observed. However, because we were not measuring denitrification directly, we could not determine whether denitrification in mid-summer was very low or if it was masked by ammonium desorption from the sediments.
Approach: Long term measurements - We will measure benthic fluxes twice a year at the upper and mid estuarine sites, at the beginning and end of the summer. We will look for changes in fluxes as the salinity structure changes through natural variations in river discharge and sea level. To separate the effects of salinity from organic matter inputs (see above) we will measure exchangeable pools of nutrients in the sediments. Process studies - In laboratory experiments we will compare benthic fluxes in cores with a range of salinities. Cores from our upper and mid-estuarine sites will be collected in early spring and fluxes will be measured before and after the salinity of the overlying water is increased to typical end-of-summer values. Fluxes will be measured several times over a six week period. Companion cores will be taken and treated the same way and occasionally sacrificed for measurements of porewater and exchangeable ammonium. We are currently seeking Sea Grant funding to directly measure denitrification under different salinity regimes both in the lab and field using the isotope pairing technique (Nielsen 1992).
QUESTION 5. How does the structure and function of higher trophic levels respond to variations in organic matter, nutrients and water fluxes? (Q5 in Conceptual Model, pg 2-4)
RATIONALE - Estuarine biota are profoundly affected by changes to the spatial and temporal availability of habitat and food (Kennedy 1990). Fish distribution, production, and habitat choice at both the microhabitat and whole-estuary scale are influenced by the physicochemical environment (salinity, temperature, DO, water flux), food availability, competitive interactions and predators (Livingston et al. 1997, Pennock et al. 1994, Drinkwater and Frank 1994, Keller et al. 1990, Nixon et al. 1986, Deegan et al. 1986). Access by fish to the highly productive intertidal marsh habitat (Kneib 1994) varies with duration, frequency, and depth of marsh flooding (Q2, pg. 2-10). By altering the predator field, habitat availability, and food sources (Deegan 1990, Cowan et al. 1993), river discharge can change the location of peak fish production. One way this occurs is the stimulation of plankton production by nutrients in increased river flow which in turn favors the production of pelagic over benthic species (Livingston et al. 1997, Deegan et al. 1994, Nixon et al. 1986). Sources of organic matter supporting higher trophic levels vary along the estuary: river inputs of POC dominate in the upper estuary while salt marsh and phytoplankton inputs dominate in the lower estuary (Deegan and Garritt 1997). To deal with this complexity, Allen and Hoekstra (1992) and Brandt and Mason (1994) suggest using spatially explicit models to link the biological and physical attributes of the habitat and to predict individual and system-wide fish production in response to changing conditions.
The functional roles of higher trophic levels may also change with variations in organic matter, nutrients and water flux. For example, phytoplankton cropping by bivalves which helps prevent blooms and periodic oxygen depletion occurs only where water residence time is less than the clearance time of bivalves (Smaal and Prins 1993, Dame 1996, Cloern 1996). Changing either water residence time or bivalve population levels will alter this balance. Sullivan et al. (1991) suggested that control of the pelagos by the benthos varies with stratification, again indicating the interaction between hydrodynamics and biotic function. Deegan (1993) has shown that the balance between growth and mortality determines whether fish are net transporters of material; growth and mortality are controlled by water flux and food availability (Cadigan and Fell 1985, Kneib 1986)
Specific Research Questions: 5a) How does the production of higher trophic levels vary spatially and temporally with changes in organic matter, nutrients and water flux?
During 1993/94 we measured an increase in total fish abundance,
and a 10-fold increase in the abundance of Fundulus heteroclitus
(mummichog) and Menidia menidia (Atlantic silversides) compared
to 1964 (Fig 18). These two species account for almost 70% of the
biomass and are important food for piscivores. The cause of this increase
is unknown but could be due to higher river discharge and sea level, lower
salinity, and fewer predators because of exclusion of stenohaline fish
and long-term declines in striped bass (Deegan et al., in prep). We do
not know if we have observed a trend or simple year-to-year variation because
long-term records of fish abundance are simply not available for northern
estuaries.
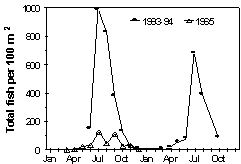
Approach: We will use long-term measurements of fish communities in Plum Island, Wells, and North Inlet, process studies of growth and mortality, and modeling to integrate the effects of multiple factors on fish production. We will annually measure fish abundance at 5 sites in different regions of Plum Island Sound. Three sites will be located in the upper- (riverine dominated), mid- (marsh dominated) and lower- (ocean dominated) estuary. We will also contrast annual fish abundance in the Rowley River (high DIN) to that of the Parker River (low DIN). We will use traditional techniques (seining and trawling) supplemented with acoustic surveys. Across estuaries and sub-estuaries we will relate annual estimates of fish abundance to river discharge, nutrient and organic matter delivery, sea level and abundance of top predators. For example, we hypothesize that regionally increased striped bass abundance will cause declines in forage fish.
We will determine the importance of the marsh for feeding and protection from predators by conducting growth and predator exclusion experiments with cages and examining gut contents (Murphy 1991, Kneib 1994). We will determine fish abundance and height of flooding requirements, and the importance of geomorphology (esp. drainage density and edge) using flume nets (Murphy 1991). This will be combined with the extent, duration and depth of flooding (Q2, pg. 2-10) to predict fish production. Because the marshes in the Wells Reserve are sinking relative to sea level (Kelley et al. 1993), they should be more accessible (flood deeper, more frequently and longer; Murphy 1991) and produce more fish than Plum Island marshes.
We will construct spatially explicit models of potential fish production linking measures of habitat suitability (Fig 19) to bioenergetic or individual based growth models (e.g., Cowan et al. 1993, Brandt and Mason 1995). These models will integrate the interannual variability of several factors that determine total fish production.
5b) How does the partitioning of production between benthic and pelagic fish change with variations in organic matter and nutrients?
Our trophic model (Deegan et al. 1994) predicts that total production and partitioning between benthic and pelagic fish will depend on the relative abundance and availability of high quality phytoplankton and low-quality marsh or terrestrial organic matter. The benthic estuarine food web is more dependent on marsh production (Deegan and Garritt 1997). This suggests that variations in terrestrial, marsh or algal production may influence the quantity and type (benthic, pelagic, herbivore, carnivore) of fish produced. We predict that benthic fish will decrease with reduced inputs of marsh organic matter and pelagic fish will increase with increased DIN loading because of the efficient pelagic grazing pathway (Willemsen 1980, Deegan et al. 1994).
Approach: We will use long-term measurements in
experiments and sites within estuaries, and mesocosm experiments to determine
the effects of organic matter on fish production. We will monitor the production
and d 13C, d
15N and d34S
values of fish and invertebrates in the marsh creek fertilization and detritus
removal experiments. The fish community will be monitored using fyke/flume
nets (Rountree and Able 1993). Production of Fundulus and Menidia
will
be estimated based on growth (otoliths: Secor et al. 1995, Barkman et al.
1981) and mortality (length- frequency and abundance: Ricker 1975).
We will examine annual variations in fish abundance at the 5 sites in Plum Island, and in Wells and North Inlet for differences in benthic and pelagic fish production related to organic matter-nutrient loadings. Zooplankton, benthic invertebrates (Q4, pg. 2-15) and Fundulus and Menidia d 13C, d 15N and d34S values will help determine if the differences in community structure correlate with differences in organic matter inputs.
In mesocosms with known inputs of DIN and organic matter and intact benthos, we will measure the production of pelagic and benthic fish. We will run these mesocosms with the same nutrient & organic matter loading conditions as the pelgic mesocosms described in Q3 (pg. 2-12) and measure the biomass of phytoplankton, zooplankton, macrobenthos, and fishes as well as identify the source of food web organic matter with stable isotopes (Results from Prior).
5c) What is the role of higher trophic levels in regulating trophic structure and in transporting carbon and nitrogen across estuarine interfaces?
Plum Island Sound is well known for the production of soft-shelled clams, quahogs and mussels (>14,000 bushels, 1994; Div. of Marine Fisheries). The mid-estuarine dip in phytoplankton biomass and peak in nutrients (Vorosmarty and Loder 1994) corresponds to one of the largest and most productive Mya beds indicating they may be important in controling phytoplankton and regenerating nutrients. Interannual variation in forage fish, especially Menidia, suggests that their importance in transporting energy and nutrients may vary with estuarine conditions.
Approach: In the upper and mid-estuary, where water residence time is long and Mya densities are greatest, we anticipate grazer control of phytoplankton. Near the mouth of the estuary, water residence time is short and we expect hydrologic control. Comparison of years with high and low river discharge will also indicate how controls on phytoplankton vary. In a year with high river discharge we expect the plankton to be under hydrologic (residence time), not grazer control. We will monitor annually the size structure, density and extent of bivalve beds and couple this with models of nutrient regeneration and filter-feeding capacity (Dame 1996) to determine bivalve importance in controlling ecosystem processes.
We will measure the importance of fishes in transporting nutrients and energy across ecosystems boundaries by annually measuring the abundance of Menidia and Fundulus entering and leaving saltmarshes and the estuary. Comparison of the creek fertilization experiment, the detritus removal experiment and control creeks over several years should indicate the relative importance of organic matter sources versus water flux and habitat availability (marsh flooding) in determining net export.
RESEARCH APPROACH
This is an integrated program consisting of long and short-term experiments, long-term field measurements, comparative ecosystem studies and modeling. Here we describe long-term experiments that will aid in understanding the mechanisms controlling key ecosystem processes, long-term field measurements that will form our core datasets, comparative research to be conducted in our parallel sites, and modeling plans to scale to larger regions. The schedule of experiments is given on page 4-2. Long term measurements are summarized in Table 2.
Our proposed research addresses each of the 5 LTER core areas. 1) We will measure temporal and spatial patterns of marsh and estuarine primary production and the effect of N+P fertilization on marsh production. 2) We will devote considerable time monitoring temporal and spatial patterns of bacteria, zooplankton, fish and shellfish. 3) Inputs, stores and dynamics of organic matter will be investigated in the watershed, marshes, and estuarine waters. 4) Nutrient inputs from the watershed will be measured and movement through the ecosystem will be traced. 5) Finally, our research focuses on the effects of changes in climate, sea level and land cover on trophic dynamics, thus addressing the issue of disturbance.
LONG TERM EXPERIMENTS: Eutrophication of coastal systems from additions of nitrogen is arguably the principal and most pervasive anthropogenic alteration to coastal ecosystems around the globe. While the effects of eutrophication of coastal waters are widely studied, less is known about the response of coastal marshes to enhanced N loading. Marshes exist in a fragile dynamic balance between accretion and isostatic sea level rise and processes that affect rates of accretion can alter that balance. N loading can affect accretion by altering macrophyte productivity, allocation to above or below ground biomass, and organic matter decomposition. We will establish 2 marsh fertilization experiments and a detrital removal experiment to examine the effects of these alterations on primary production, organic matter accumulation/decomposition and biotic community structure and production.
Direct Marsh Fertilization: Fertilizer will be applied monthly during the growing season to replicated marsh swaths in both a brackish and a salt marsh site along the Parker River. Commercial fertilizer (16NH4+-N, 1PO43-P) will be applied at low tide at the rate of 100 kg-N ha-1yr-1. Swaths will be 10 m wide, include tidal creeks and extend from creek bank, across the high marsh to the uplands. This design will provide uniform loading to the entire elevational gradient. Fertilization is ongoing at North Inlet (Morris) and will be initiated at Wells.
Tidal Creek Fertilization of Marsh: There are a number of sites in Plum Island where marshes are connected to the open water by culverts. Replicated marsh sites will be fertilized by dripping N-P fertilizer directly into the creek at the culverts during flood tide. Dissolved N and P fertilizer will be added in sufficient quantity to raise DIN concentration to 100 µM and P to 6.25 µM. We expect this type of application to yield significantly different results than the direct fertilization because the loading rate will vary in proportion to the areal extent of marsh flooded with each tide. This type of application closely resembles what happens to marshes as inorganic nutrient concentrations in tidal waters increase.
Detritus Removal: Approximately 35 ha of Plum Island Sound intertidal high marsh dominated by Spartina patens is regularly hayed by commercial farmers. While this process used to be common and extensive in New England marshes, the operation in Plum Island Sound is one of the last. Haying removes >90% of annual aboveground production and biomass. This represents a significant loss of nutrients (Knapp and Seastedt 1983) and organic matter that would otherwise be available for marsh detritivores and microbes, export or peat accumulation.
Experimental Measurements: At control and experimental sites we will measure: tide height, plant community structure, macrophyte and microalgal production, community respiration, marsh elevation and accretion, sediment and biomass nutrient and organic matter content, porewater chemistry, marsh water table depth, invertebrate populations (amphipods, harpacticoids, ribbed mussel), fish populations (Menidia and Fundulus), and organism stable isotope ratios. Areas will be monitored for 3 years prior to manipulation. Sampling frequency will be high the first 2 years and then decrease as scales of variability are identified.
COMPARATIVE ECOSYSTEM RESEARCH: We seek conclusions and models that are applicable across a wide range of coastal waters and ecosystems. We have formally incorporated two National Estuarine Research Reserves (North Inlet, SC and Wells, ME) into our proposed LTER program. Many parallel measurements and 2 experiments will be conducted at these sites. Current NOAA funding to these sites will support this activity which thus leverages NSF LTER funding.
Comparative ecosystem work continues to be a major activity of our LMER. We have led three LMER working group intercomparisons of 1) riverine organic matter quality and lability (Hopkinson et al. submitted), 2) stable isotopic analysis of organic matter sources fueling higher trophic levels (Deegan et al. in prep), and 3) watershed hydrology and nutrient loading (Finn chair). The N-15 addition experiment planned for the Ipswich River will enable direct comparison with the N-15 streams study (LINX) led by Webster et al. Likewise, detritus removal experiments conducted at other LTERs will provide interesting contrasts to our haying experiments. We expect to develop additional intercomparisons and partnerships within the LTER and coastal research communities.
INTERNATIONAL LTER: Hopkinson, Deegan and Peterson have been participating in an NSF funded project of Ken Tenores (UMd) that is aiding Portuguese scientists develop a land-margin ecosystem research program similar to that in the U.S.
REGIONAL MODELING COLLABORATIONS: We are collaborating with C. Driscoll of the Hubbard Brook LTER on a model of nutrient dynamics in the Merrimac River watershed and with C. Vorosmarty and colleagues at the Univ. of New Hampshire on a regional GIS-based model of water, nutrient and organic matter flux from the Gulf of Maine watershed to the sea
OUTREACH, EDUCATION, LAY COLLABORATIONS: The principal investigators work with Mass. Audubon, the Essex County Greenbelt Association, the Ipswich Watershed Association and the Mass. Division of Environmental Protection and Coastal Zone Management to apply our knowledge to address environmental issues of public concern. The U.S. Fish and Wildlife is building an environmental education center and headquarters at Plum Island and we will periodically provide posters and displays to them and to the Reserve headquarters at Wells and North Inlet. We also share information through our Plum Island web site which includes a summary of research goals, maps, data and links to other potentially interesting sites.
SYNTHESIS. Simulation modeling and data analysis will integrate research conducted at a variety of temporal and spatial scales to address the responses of ecosystems at the land-sea interface to long term changes in watershed land cover, climate and sea level. (Q6 in Conceptual Model, pg 2-4)
RATIONALE - To identify and understand long-term changes and fundamental ecosystem processes, our approach includes long term field measurements, short and long-term experimentation, and comparative ecosystem studies. Although the core of our research is experimental, significant effort will be expended on developing and improving models to: 1) provide a succinct summary of our current understanding of land margin ecosystems, 2) rigorously test our understanding of ecosystem processes by comparing model predictions to observations, especially across sites, 3) identify processes or areas of research that require further experimental investigation, and 4) extrapolate in space and time to examine most probable ecosystem response to perturbations caused by changes in watershed land cover, climate and sea level.
Hydrologic - hydrodynamic integration. Transport processes in estuaries have a critical role in determining the location of pelagic blooms, the duration and extent of marsh flooding, environmental conditions at the sediment-water interface, and conveyance of materials. In turn, estuarine transport is driven by freshwater input, tidal forcing, wind stress, thermal balance, and the geomorphology of the estuary. Of these drivers, freshwater discharge is of particular importance, since the timing and magnitude of discharge can strongly influence ecosystem function and is often either directly or indirectly under Mans influence (Hopkinson and Vallino 1995, Dynesius and Nilsson 1994). Consequently, improving our estuarine transport model (Vallino and Hopkinson 1997) and linking it to our watershed hydrology model is a primary synthesis focus.
The duration, frequency, and extent of marsh flooding determines the strength of exchange between marsh and estuarine water which directly impacts marsh productivity by altering marsh salinity. We plan to extend our advection-dispersion (AD) model (Vallino and Hopkinson 1997) to account for spring-neap tidal variations in a manner similar to the Vörösmarty and Loder (1994) model. The AD model will be developed in two dimensions (latitude and longitude), and marsh elevation will be accurately surveyed so that anomalies in mean sea level will be translatable into changes in marsh flooding extent. We will also develop a tidally explicit 1D or 2D hydrodynamic model using shallow water hydrodynamic equations (Foremanet al., 1993). This model will largely be used to interpret field observations that are taken in conjunction with marsh experiments, such as the planned marsh fertilization experiments (see Research Questions 2 & 5).
We will couple our estuarine transport models to watersheds by driving them with output from the river routing and processing model (see Q1). We will test our coupled hydrologic-hydrodynamic model by comparing predictions of freshwater discharge, salinity distributions, current velocities, and tidal heights to observations.
Synthesis of effect of land use, climate and sea level change on estuarine food webs. The dynamics of estuarine food webs are affected by the quantity and quality of organic matter inputs as well as the level of DIN inputs. To understand how organic matter and DIN resources are utilized by the microbial food web and how the resulting dynamics of the microbial consortium are propagated to higher trophic levels, we have proposed several micro- and mesocosm experiments (see Research Questions 3, 4, and 5). So that understanding gained from these experiments can be used to predict how estuarine systems will respond to changes in land use, climate and sea level, we will develop estuarine food web models with an emphasis on OM processing and linkages to higher trophic levels.
Under our current LMER project, we developed a generalized estuarine model with highly aggregated benthic and pelagic compartments (Hopkinson and Vallino 1995) (also see http://eco37.mbl.edu/adv_disp.htm). In order to improve this model, we plan to increase resolution in benthic remineralization and incorporate marsh-water coupling. We will also increase the breadth of pelagic organisms so that model output can be directly compared to experimental observations and better linked to higher trophic levels.
Our modeling approach involves three phases of development: 1) identification of key state variables and their interconnection, 2) model refinement and parameter estimation, and 3) comparison of model predictions to field observations by embedding the food web into the AD transport model. In phase 1 of development, identification of state variables and interconnections will be based on modeling transformations of inorganic nutrients and organic matter into higher trophic levels. In phase 2, we have been using mesocosm experiments (see Research Questions 3, 4, and 5) coupled to data assimilation techniques (Crispi and Mosetti 1993, Marcos and Payre 1988) to improve model structure and estimate model parameters. Techniques to simplify models yet retain essential dynamics (such as aggregation (Warwick 1989), time delays (Gibilaro and Lees 1969), time scale separation (Maas and Pope 1992)) will be a strong focus of this part of model development. Phase 3 involves rigorously testing the model to field observation by accounting for transport and inputs from watershed and ocean. Once sufficient accuracy is achieved by the coupled watershed-estuarine model, scenarios will be run to examine system response to perturbations such as land use, climate, and sea level change.
Our first models of higher trophic levels (i.e., pelagic and benthic fish) will use standard bioenergetic approaches (Ney 1993); however, these models inherently assume that higher trophic level organisms can be modeled as continuously distributed. In reality, many organisms are discretely distributed (patchy) and exhibit complex behavior such as schooling. Modeling patchy distributions of organisms requires a different modeling approach, such as individual (Letcher et al. 1996) or particle-type models (Zhou and Huntley 1996). Although these models will require significant more development, we foresee using the AD model coupled to a lower trophic-level food web model to generate a 3D environment of food resources, temperature, oxygen concentration, salinity, and intertidal marsh connectivity (see Research Question 2). Within these modeled "habitats" an individual based model of higher trophic level organisms would operate. Acoustic measurements would be used to compare model predictions to observations (Goyke and Brandt 1993).
Synthesis of effect of land use, climate and sea level change on patterns of estuarine ecosystem metabolism. Processes governing the balance between autotrophy and heterotrophy determine net ecosystem productivity of estuaries and the magnitude of organic carbon and inorganic nitrogen transported to the coastal zone (Hopkinson and Vallino 1995). We plan to improve our model of whole estuarine metabolism to examine those processes which may alter net ecosystem productivity.
To place focus directly on biogeochemical transformations (e.g., oxygen uptake and nitrification) rather than organisms, as in food web models, organisms can be replaced by reactions representing fundamental biochemical transformations, where the state variables of the model become reaction extents and substrate concentrations. To obtain a solution, an optimization problem is constructed in which a cost function (such as energy throughput) is maximized subject to constraints on energetics, electron balances, substrate utilization kinetics, and mass. We have developed such a model to examine bacterial growth (Vallino et al.1996), and plan to develop a similar model, without biotic compartments, for estuarine metabolism. Measurements of whole system metabolism (Balsis et al.1995), as determined from diel changes in water column oxygen concentration, will be used to develop and test the optimization model.
Synthesis - Improved predictions of estuarine fisheries production based on a N economy and incorporating information on land use, organic matter loading, DIN loading, sea level variation and habitat.
We may be able to understand better the relationships between fisheries yield and freshwater discharge if we include new information on DIN, dissolved and particulate organic matter as well as physical characteristics of the estuaries. In contrast to the synthetic efforts on lake ecosystems, little comparative work has been done on the production of higher trophic levels in estuaries. Comparative databases on estuaries (Nixon 1981, Boynton et al. 1982, Deegan et al. 1986, 1997) have laid the foundation for considering the importance of various factors influencing fisheries production. We will extend the analysis of Deegan et al. (1986) and include new information on dissolved and particulate organic matter as well as DIN loading. We will initially develop this model based on detailed information on land cover relationships, habitat availability and fish production in Plum Island estuary and our comparative, Wells and North Inlet sites for which we have detailed information. This will allow us to look for differences in fisheries response to freshwater discharge which may be related to biogeographic patterns. We may be able to make estimates of past fisheries yield using information from land cover export relationships, old land use data and our new statistical models. We can compare these estimates to historical estimates of fisheries yield (adjusted for fishing effort).
Table 2. Long term site core measurements to be conducted at regular
frequencies in watersheds, marshes and tidal waters. Notes: P=Plum Island,
W=Wells, N=North Inlet; Core: 1) primary production, 2) dynamics of populations,
3) organic matter, 4) nutrients, 5) disturbance.
| LOCATION | CORE | VARIABLES | METHOD | ESTUARY - # SITES | FREQUENCY |
| AIRSHED | 5 | Wet/Dry temperature (T), insolation, precip, wet & dry N/P deposition | Campbell, LiCor, | P - 3 (coast-inland); W-1, N-1 | Hourly |
| 5 | Temperature, precipitation | US Weather Service | 5 sites in and around watershed | Hourly to daily | |
| WATERSHED | |||||
|
|
3,4,5 | Temp, H2O level, NH4+, NO3-, PO43-, DO(N,P,&C) PO(C&N), TSS | Isco autosampler and logger, Alpkem autoanalyzer, UV& high temp oxidation, CHN, gravimetric | P - 3 sites representing range of land covers | Weekly with special storm event sampling |
|
|
3,4 | Sedimentation mineral and organic depth and mass | Marker horizon depth, plate accumulation, CHN, gravimetric | P - 5 along continuum | Quarterly |
|
|
3,4 | Temp, NH4+, NO3-, PO43-, DO(N,P,&C) PO(C&N), TSS, alk, pH | As above | Ipswich and Parker Dams | Weekly with special storm event sampling |
| 5 | Discharge | USGS station | Ipswich and Parker Dams | Hourly | |
| ESTUARINE
MARSH |
1 | Macrophyte productivity | Phenometric | P-3 marsh, 2 direct marsh, 2 creek fertilization, 2 haying sites; W-1, N-3 | P&W-Bimonthly- grow season; N-mo |
| 1 | Algal biomass | Pigment analysis | As above | Monthly - grow season | |
| 3,4 | Accretion | SET tables marker horizons, sedimentation plates | As above | Quarterly | |
| 3,4 | Organic and ash content | Gravimetric, CHN | As above | Yearly | |
| 2,3 | Sediment respiration | IRGA - custom chamber | As above | Yearly | |
| 3,4,5 | Porewater - NH4+, NO3-,PO43-, DO(C&N) Eh, pH, S2-, conductivity | Diffusion samplers | As above | Monthly - bimonthly | |
| 5 | Groundwater height | Pressure transducers | P - 3 marshes, 5/transect, W-1 transect, N-1 transect | Hourly | |
| 2 | Birds | direct counts | P, W, many sites | bi-weekly by Audubon and USFWS | |
| ESTUARINE
WATERBODY |
1,2,3,4,5 | Light, DO, T, cond, NH4+, NO3, PO43, DO(N,P,C), PO(C,N), TSS, chl, DO2, Prim. Prod &Resp., C&N stable isotopes (DOM, POM, DIC), DO14C, DI14C, bacteria, zooplankton | Licor, Hydrolab, autoanalyzer, UV & high temp oxidation, CHN, gravimetric, acetone /fluorescence, O2, DIC, IRMS, direct count, net tows | P-1 (transect),
W-1 (no isotopes) N-3 (no isotopes) |
P quarterly at standard tide, W-4, N-12/d every 20 days |
| 5 | Temp, DO, conductivity, depth | Hydrolab, autosamplers | P-2 (upper and lower), W-4, N-3 | 0.5 hr | |
| 3,4 | Benthic fluxes | Core incubations | P-2 (upper and mid) | Semi-annually | |
| 2,3 | Mya, Geukensia, Fundulus and Menidia | Stable isotopes - IRMS | P-5, W-1 | Yearly | |
| 2,3 | Fundulus & Menidia dynamics | Seine, mark-recapture | P-1 | Yearly | |
| 2,3 | Fish | Seine, trawl, acoustics | P-5, W-1, N-3 | Monthly - season | |
| 2,3 | Shellfish landings | Shellfish warden reports | P, W | daily |
LITERATURE CITED
Alderman, D., B. Balsis, I.D. Buffam, R.H. Garritt, C.S. Hopkinson and J.J. Vallino. 1995. Pelagic metabolism in the Parker River/Plum Island Sound Estuarine System. Biol. Bull. 189: 250-251.*
Allen, T.F.H. and T.W. Hoekstra. 1992. Towards a Unified Ecology. Columbia University Press. New York.
Aller, R. 1994. Bioturbation and remineralization of sedimentary organic matter: effects of redox oscillation. Chem. Geol. 114: 331-345
Armstrong, N. 1982. Responses of Texas estuaries to freshwater inflows, pages 103-120. In: V. Kennedy (ed.), Estuarine Comparisons. Academic Press.
Ayvazian, S. G., L. A. Deegan and J. T. Finn. 1992 Comparison of nekton use of estuarine habitats across two biogeographic zones. Estuaries 15: 368-393.
Balsis, B., D. Alderman, I.D. Buffam, R.H. Garritt, C.S. Hopkinson and J.J. Vallino. 1995. Total system metabolism in the Plum Island Sound estuary. Biol. Bull. 189: 252-254.*
Barkman, RC; Bengtson, DA; Beck, AD. 1981. Daily Growth of the Juvenile Fish (Menidia menidia) in the Natural Habitat Compared With Juveniles Reared in the Laboratory. RAPP. P.-V. REUN. CIEM., vol. 178: 324-326.
Bencala, K. and R. Walters. 1983. Simulation of solute transport an a mountain pool-and-riffle stream: a transcient storage model. Water Resources Research. 19: 718- 724.
Billen, G., C. Lancelot and M. Meybeck. 1991. N,P and Si retention along the continuum from land to ocean. In: Mantoura, R.F.C., J. -M. Martin and R wollast (eds.), Ocean Margin Processes in Global Change, pp. 19-44. John Wiley, New York.
Boumans, R.M. and J.W. Day. 1994. Effects of two Louisiana marsh management plans on water and materials flux and short-term sedimentation. Wetlands. 14: 247-261.
Boumans, R.M.J. and J.W. Day, Jr. 1993. High precision measurements of sediment elevation in shallow coastal areas using a sedimentation-erosion table. Estuaries 16: 375-380.
Boynton, W., M. Kemp and C. Keefe. 1982. A comparative analysis of nutrients and other factors influencing estuarine phytoplankton production, pp. 69-90. In: V. Kennedy (ed.), Estuarine Comparison. Academic.
Boynton, W.R. and W.M. Kemp. 1985. Nutrient regeneration and oxygen consumption by sediments along an estuarine salinity gradient. Mar. Ecol. Prog. Ser. 23: 45-55.
Brandt, S. B. and D. M. Mason. 1994. Landscape approaches for assessing spatial patterns in fish foraging and growth. Landscape appraoches for assessing spatial patterns in fish foraging and growth. Pp 211- 241 in: D. Strouder, K. Fresh, R. Feller (eds.) Theory and application in fish feeding ecology. Univ. South Carolina Press.
Briggs, J. C. 1974. Northern hemisphere cold-temperate and arctic regions, pp. 275-278. In: Marine Zoogeography. McGraw-Hill Book Company.
Bronk, D. A. and P. M. Glibert. 1991. A 15N tracer method for the measurement of dissolved organic nitrogen release by phytoplankton. Mar. Ecol. Prog. Ser. 77: 171-182.
Bronk, D. and P. Glibert. 1993. Application of a 15N tracer method to the study of dissolved organic nitrogen uptake during spring and summer in Chesapeake Bay. Mar. Biol. 115: 501-508.
Buchsbaum, R.N. 1996. Resource protection in the Plum Island Sound/Rivers Ecosystem. Final Project Report of the Plum Island Sound Minibay Project of the Massachusetts Bay Program.*
Cadigan, K.M. and P.E. Fell. 1985. Reproduction , growth and feeding habitats of Menidia menidia ina tidal-marsh estuarine system in southern New England. Copeia 1: 21-26.
Cahoon, D.R. 1994. Recent accretion in two managed marsh impoundments in coastal Louisiana. Ecol.Appl. 4: 166-176.
Cahoon, D.R. and D.J. Reed. 1995. The relationship between marsh surface topography, hydroperiod, and soil accretion in a deteriorating Louisiana salt marsh. J. Coastal Res. 11:357-369.
Cahoon, D.R. and R.E. Turner. 1989. Accretion and canal impacts in a rapidly subsiding wetland. II. Feldspar marker horizon technique. Estuaries 12: 260-268.
Cahoon, D.R., D.J. Reed and J. Day. 1995. Estimating shallow subsidence in microtidal salt marshes of the southeastern United States: Kaye and Barghoorn revisited. Mar. Geol. 128: 1-9.
Callaway, J.C., R.D. DeLaune, and W.H. Patrick. 1996. Chernobyl 137Cs used to determine sediment accretion rates at selected northern European coastal wetlands. Limnol. Oceanogr. 41: 444-450.
Cammen L. M. 1980. The significance of microbial carbon in the nutrition of the deposit feeding polycheate Nereis succinea. Mar. Biol. 61: 9-20.
Carini, S.S., N.B.Weston, C.S. Hopkinson, J. Tucker, A.E. Giblin and J.J. Vallino. 1996. Gas exchange rates in the Parker River estuary, Massachusetts. Biol. Bull. 191:333-334.*
Carlozzi, C., K. King and W. F. Newbold, Jr. 1975. Ecosystems and Resources of the Massachusetts Coast. MA Coastal Zone Management Program, Executive Office of Environmental Affairs, Boston. 68 pp.
Cloern, J. 1996. Phytoplankton dynamics in caostal ecosytems: a review with some general lessons from sustained investigation of San Fransico Bay, California. Rev. Geophys. 34: 127-168.
Coffin, R. B., B. Fry, B. J. Peterson and R. T. Wright. 1989. Carbon isotope compositions of estuarine bacteria. Limnol. Oceanogr. 34: 1305-1310.
Cowan, JH Jr; Rose, KA; Rutherford, ES; Houde, ED. 1993. Individual-based model of young-of-the-year striped bass population dynamics. 2. Factors affecting recruitment in the Potomac River, Maryland. TRANS. AM. FISH. SOC. 122: 439-458.
Crawford, C. C., J. E. Hobbie and K. L. Webb. 1973. The utilization of dissolved free amino acids by estuarine microorganisms. Ecology 55: 331-363.
Crispi, G. and Mosetti, R. (1993) Adjoint estimation of aquatic ecosystem parameters. COENOSES 8, 11-14.
Currie, D.J. and J. Kalff. 1984. Can bacteria outcompete phytoplankton for phosphorus? Microbial Ecology 10: 205-216.
DElia, C.F., J.G. Sanders, and W.R. Boynton. 1986. Nutrient enrichment studies in a coastal plain estuary: Phytoplankton growth in large-scale, continuous cultures. Canadian J. Fish. Aquatic Sci. 43: 397-406.
Dame, R. F. 1996. Ecology of marine bivalves An ecosystem approach. CRC Press, Boca Raton. 254 pp.
de Jonge, V. N. and L. A. Villerius. 1989. Possible role of carbonate dissolution in estuarine phosphate dynamics. Limnol. Oceanogr. 34(2): 332-340.
Deegan, L. 1993. Nutrient and energy transport between estuaries and coastal marine ecosystems by fish migration. Can. J. Fish. Aq. Sci. 50:74-79.
Deegan, L. 1990. Effects of estuarine environmental conditions on populations dynamics of young-of-the-year gulf menhaden. Marine Ecology Progress Series 68:195-205.
Deegan, L. A., J. T. Finn, S. G. Ayvazian and H. E. Geyer. The influence of macroalgae in an eelgrass ecosystem on fish and decapod production. Submitted to Mar. Ecol. Prog. Ser.
Deegan, L.A. 1993. Nutrient and energy transport between estuaries and coastal marine ecosystems by fish migration. Canadian J. Fish. Aquatic Sci. 50: 74-79.*
Deegan, L.A. and Garritt, R.H. 1997. Evidence for spatial variability in estuarine food webs. Mar. Ecol. Prog. Ser. 147: 31-47.*
Deegan, L.A. and R.N. Buchsbaum. 1996. The effect of habitat loss and degradation on fisheries. In: Buchsbaum, R.N., W.E. Robinson, and J. Pederson (eds). The decline of fisheries resources in New England: Evaluating the impact of overfishing, contamination, and habitat degradation. Massachusetts Bays Program, University of Massachusetts Press.*
Deegan, L.A., J. Finn, C.S. Hopkinson, B.J. Peterson, A.E. Giblin, B. Fry and J. Hobbie. 1994. Flow model analysis of the effects of organic matter-nutrient interactions on estuarine trophic dynamics. In- Dyer, K. and R. Orth (eds.), International Symposium Series, Changes in fluxes in estuaries: implications from science to management. Olsen and Olsen Publ., Fredensborg, DK. pages 273-281.*
Deegan, L.A., J.T. Finn, S.G. Ayvazian, C.A. Ryder and J. Buonaccorsi. 1997. Development and validation of an estuarine biotic integrity index. Estuaries. Accepted.*
Deegan. L. A., J. Day, J. Gosselink, A. Yáñez -Arancibia, G. Soberon-Chavez and P. Sanchez-Gil. 1986. Relationships among physical characteristics, vegetation distribution and fisheries yield in Gulf of Mexico estuaries, pp. 83-100. In: D. Wolfe (ed.), Estuarine Variability. Academic Press.
Drinkwater, K.F. and K.T. Frank. 1994. Effects of river regulation and diversion on marine fish and invertebrates. Aquatic Conserv: Mar. Freshwater. Ecosyst. 4: 135-151.
Dynesius, M. and Nilsson, C. (1994) Fragmentation and flow regulation of river systems in the northern third of the world. Science 266: 753-762.
Federer, C. A. and D. Lash. 1978. BROOK: A Hydrologic Simulation Model for Eastern Forests. Research Report #19. Water Resources Center, University of New Hampshire, Durham, NH.
Findlay, S. and K. Tenore. 1982. Nitrogen source for a detritivore: Detritus substrate versus associated microbes. Science 218: 371-372.
Finn, J.T, and C.S. Hopkinson. 1997. Organic matter loading to estuaries from upland watersheds. Report to Massachusetts Water Resources Research Center. pp. 63.
Foreman, M.G.G., Henry, R.F., Walters, R.A., and Ballantyne, V.A. (1993) A finite element model for tides and resonance along the north coast of British Columia. J. Geophys. Res. 98: 2509-2531.
Fox, L. E., S. L. Sager, and S. T. Wofsy. 1986. The chemical control of soluble phosphorus in the Amazon estuary. Geochim. Cosmochim. Acta 50: 783-794.
Froelich, P. N. 1988. Kinetic control of dissolved phosphate in natural rivers and estuaries: A primer on the phosphate buffer mechanism. Limnol. Oceanogr. 33(2): 649-668.
Fry, B., C.S. Hopkinson, A.L. Nolin and S. Wainright. 13C/12C composition of marine dissolved organic carbon. Submitted Geol. Soc. Am. Bull.*
Fry, B., C.S. Hopkinson, A.L. Nolin, B. Norrman and U. Zweifel. 1996. Long term decomposition of DOC from experimental diatom blooms. Limnol. Oceanogr. 41: 1344-1347.*
Fry, B., E. Peltzer, C.S. Hopkinson, A.L. Nolin and L. Redmond. 1996. Analysis of marine DOC using a reference dry combustion method. Mar. Chem. 54: 191-201.*
Fry, B., M.A.J. Hullar, B.J. Peterson, S. Saupe and R.T. Wright. 1992. DOC production in a salt marsh estuary. Arch. Hydrobiol. Beih. 37: 1-8.
Fry, B., S. Saupe, M.A.J. Hullar and B. J. Peterson. 1993. Platinum-catalyzed combustion of DOC in sealed tubes for stable isotopic analysis. Mar. Chem. 41: 187-193.*
Gardner, W.S., S.P. Seitzinger and J.M. Malczyk. 1991. The effects of sea salts on the forms of nitrogen released from estuarine and freshwater sediments: Does ion pairing affect ammonium flux? Estuaries 14:157-166
Geyer, W., D. Jay, F. Prahl, J. Morris and E. Turner. 1997. The coupling of physics, biogeochemistry and ecology. SCOPE Working Group Report. In- J. Hobbie (ed.), Estuarine Synthesis: the next decade. Woods Hole, MA. (in prep).
Gibilaro, L.G. and Lees, F.P. (1969) The reduction of complex transfer function models to simple models using the method of moments. Chem. Eng. Sci. 24, 85-93.
Goldman, J., D. Caron and M. Dennett. 1987a. Regulation of gross growth efficiency and ammonium regeneration in bacteria by substrate C:N ratio. Limnol. Oceanogr. 32:1239-1252.
Goldman, J., D. Caron and M. Dennett. 1987b. Nutrient cycling in a microflagellate food chain: IV. Phytoplankton-microflagellate interactions. Mar. Ecol. Prog. Ser. 38: 75-87.
Goyke, A.P. and Brandt, S.B. (1993) Spatial models of salmonine growth rates in Lake Ontario. Transactions of the American Fisheries Society 122, 870-883.
Haith, D.A., R. Mandel and R.S. Wu/ 1992. GWLF: Generalized Watershed Loading Functions, Version 2.0. Department of Agricultural and Biological Engineering, Cornell University, Ithaca, NY.
Hansson, S., J.E. Hobbie, R. Elmgren, U. Larsson, B. Fry and S. Johansson. Stable nitrogen isotopes as a marker for food web interactions and fish migration a comparison of three coastal Baltic Sea stations. Ecology. In press.*
Hatcher, B., J. Imberger and S. Smith. 1987. Scaling analysis of coral reef systems: an approach to problems of scale. Coral Reefs 5:171-181.
Hayden, B. and R. Dolan. 1976. Coastal marine fauna and marine climates of the Americas. J. Biogeography 3: 71-81.
Hirshberg, D. and J. Schubel. 1979. Recent geochemical history of flood deposits in the northern Chesapeake Bay. Est. Coastal Marine Sci. 9:771-784.
Hobbie, J. E. and J. J. Cole. 1984. Response of a detrital food web to eutrophication. Bull. Mar. Sci. 35(3): 357-363.
Hobbie, J. E. and J. V. K. Helfrich III. 1988. The effect of grazing by microprotozoans on production of bacteria. Arch. Hydrobiol. Beih. Ergebn. Limnol. 31: 281-288.
Hobbie, J. E., J. J. Vallino, R. H. Garritt and I.D. Buffam. 1996. Availability of stream organic matter for estuarine biota. ASLO Abstracts, June 1996, U. Wisconsin, Milwaukee, WI.*
Hobbie, J.E. 1994. The state of the microbes: a summary of a symposium honoring Lawrence Pomeroy. Microb. Ecol. 28: 113-116.*
Hobbie, J.E., S. Johansson, U. Larsson, and B. Fry. Microbial use of DOC from land and sea. Manuscript in revision.*
Hopkinson, C. S. 1988. Patterns of organic carbon exchange between coastal ecosystems: The mass balance approach in salt marsh ecosystems, pp. 122-154. In: B-O. Jansson (ed.), Coastal-Offshore Ecosystem Interactions. Lecture Notes on Coastal and Estuarine Studies, Vol. 22. Springer-Verlag.
Hopkinson, C. S. 1992. A comparison of ecosystem dynamics in freshwater wetlands. Estuaries 15(4): 549-562.*
Hopkinson, C. S. and J.J. Vallino. 1994. Towards the development of generally-applicable models of the microbial loop in aquatic ecosystems. Microb. Ecol. 28(2): 321-326.*
Hopkinson, C. S. and J.J. Vallino. 1995. The relationship among mans activities in watersheds and estuaries: a model of runoff effects on estuarine community metabolism. Estuaries 18: 598-621.*
Hopkinson, C. S., A. E. Giblin,R.H. Garritt, J. Tucker, and M.A.J. Hullar. 1997. The influence of the benthic boundary layer on growth of pelagic bacteria. Submitted Aquatic Microb. Ecol.*
Hopkinson, C. S., B. Fry, and A.L.Nolin. 1996. Stoichiometry of dissolved organic matter dynamics on the continental shelf of the Northeastern U.S.A. Cont.Shelf Res. (in press).*
Hopkinson, C. S., B. Sherr and W. J. Wiebe. 1989. Size-fractionated coastal plankton metabolism - substrate and nutrient interactions. Mar. Ecol. Prog. Ser. 51: 155-166.
Hopkinson, C. S., L. Cifuentes, D. Burdige, S. Fitzwater, D. Hansell, S. Henrichs, P. Kähler, I. Koike, T. Walsh and B. Bergamaschi. 1993. The DON Controversy - A report. Mar. Chem. 41:23-36.*
Hopkinson, C.S, I. Buffam, J. Hobbie, J. Vallino, M. Perdue, B. Eversmeyer, F. Prahl, J. Covert, R. Hodson, M.A. Moran, E. Smith, J. Baross, B. Crump, S. Findlay, D. Foreman. 1997. Terrestrial inputs of organic matter to coastal ecosystems: an intercomparison of chemical characteristics and bioavailability. Submitted Biogeochemistry.*
Hopkinson, C.S., A.E. Giblin, J. Tucker, and R.H. Garritt. Benthic metabolism and nutrient cycling along an estuarine salinity gradient. Submitted Estuaries. *
Howarth, R. W., G. Billen, D. Swaney, A. Townsend, N. Jaworski, K. Lajtha, J.A. Downing, R. Elmgren, N. Caraco, T. Jordon, F. Berendse, J. Freney, V. Kudeyarov, P. Murdoch, and Zhao-Liang Zhu. 1996. Regional nitrogen budgets and riverine N and P fluxes for the drainages to the North Atlantic Ocean: natural and human influences. Biogeochem. 35: 75-139.
Hullar, M. A. J., B. Fry, B. J. Peterson and R. T. Wright. 1996. Microbial utilization of estuarine dissolved organic carbon: a stable isotope tracer approach tested by mass balance. Applied and Environmental Microbiology 62:2489-2493.*
Ingram, K., C. S. Hopkinson, K. Bowman, R.H. Garritt and J.J. Vallino. 1994. From watershed to estuary: assessment of nutrient loading, retention and export from the Ipswich River Basin. Biol. Bull. 187:277-278.*
Jay, D.A., R.J. Uncles, J. Largier, W.R. Geyer, J.J. Vallino and W.R. Boynton. 1997 A Review of Recent Developments in Estuarine Scalar Flux Estimation. Estuaries. 20: 262-280.*
Jerome, W. C. Jr., A. P. Chesmore and C. O. Anderson Jr. 1968. A Study of the Marine Resources of the Parker River-Plum Island Sound Estuary. Commonwealth of Massachusetts, Division of Marine Fisheries, Dept. of Natural Resources, Monograph Series #6. 79 pp.
Jones, R. I, P. J. Shaw, and H. D. Haan. 1993. Effects of dissolved humic substances on the speciation of iron and phosphate at different Ph and ionic strength. Environ. Sci. Technol. 27: 1052-1059.
Jorgensen, K. S. and J. Sorensen. 1988. Two annual maxima of nitrate reduction and denitrification in estuarine sediment (Norsminde Fjord, Denmark). Mar. Ecol. Prog. Series 48:147-154.
Joye, S.B. and J. T. Hollibaugh. 1995. Influence of sulfide inhibition of nitrification on nitrogen regeneration in sediments. Science 270: 623-625.
Keller, P., P. Doering, S. Kelly, B. Sullivan. 1990. Growth of juvenile menhaden, Brevoortia tyranus, in MERL mesocosms: effects of eutrophication. Limnol. Ocean. 35:109-122.
Kelley, J.T., D.F. Belknap and D.N. Fitzgerald. 1993. Sea level change, coastal processes, and shoreline development in northern New England. In: Field Trip guidebook for the Northeastern United States: 1993 Boston GSA. J.T. Cheney and J.C. Hepburn, eds. Contribution No. 67, Department of Geology and Geography, University of Massachusetts, Amherst, Massachusetts.
Kennedy, V.S. 1990. Anticipated effects of climate change on estuarine and coastal fisheries. Fisheries. 15: 16-25.
Kirchman, D., R. Keil, and P. Wheeler. 1989. The effect of amino acids on ammonium utilization and regeneration by heterotrophic bacteria in the subarctic Pacific. Deep-Sea Res. 36:1763-1776.
Knapp, A.K. and T.R. Seastedt. 1983. Detritus accumulation limits productivity of tallgrass prairie. Bioscience. 36: 662-668.
Kneib, R. 1994. Spatial pattern, spatial scale and feeding in fishes. pp 171-186 in: D. Strouder, K. Fresh, R. Feller (eds.) Theory and application in fish feeding ecology. Univ. South Carolina Press.
Kneib, R. T. 1986. The role of Fundulus heteroclitus in saltmarsh trophic dynamics. American Zoologist. 26:259-269.
Krone, R.B. 1985. Simulation of marsh growth under rising sea levels. In: Hydraulics and hydrology in the small computer age, edited by Waldrop, W.R. Lake Buena Vista, FL: Hydraulics Div. ASCE, p. 106-115.
Lebo, M. E. 1991. Particle-bound phosphorus along an urbanized coastal plain estuary. Marine Chemistry 34: 225-246.
Leonard, L.A. and M.E. Luther. 1995. Flow hydrodynamics in tidal marsh canopies. Limnol.Oceanogr. 40:1474-1484.
Letcher, B.H., Rice, J.A., Crowder, L.B., and Rose, K.A. (1996) Variability in survival of larval fish: disentangling components with a generlized individual-based model. Can. J. Fish. Aquat. Sci.53, 787-801.
Lewitus, A.J., E.T. Koepfler, and J.T. Morris. Trophic interactions in a salt marsh estuarine microbial food web. I. Top-down control of the phytoplankton bloom. Limnol. Oceanogr. Submitted.
Likens, G. E., F.H. Bormann, R.S. Pierce, J.S. Eaton, and N.M. Johnson. 1977. Biogeochemistry of a Forested Ecosystem. Springer Verlag, New York 146pp.
Lim, E. L., D. Caron and E. DeLong. 1996. Development and field application of a quantitative method for examining natural assemblages of protists with oligonucleotide probes. Applied and Environmental Microbiology 62:1416-1423.*
Livingston, R. J., X. Niu, F. Lewis, and G. Woodsum. 1997. Freshwater input to a gulf estuary: long-term control of trophic organization. Ecological Applications 7:277-299.
Maas, U. and Pope, S.B. (1992) Simplifying chemical kinetics: Intrinsic low-dimensional manifolds in composition space. Combustion and Flame 88, 239-264.
Mackin, J.E. and R.C. Aller. 1984. Ammonium adsorption in marine sediments. Limnol. Oceanogr. 29:250-257.
Marcos, B. and Payre, G. (1988) Parameters estimation of an aquatic biological system by the adjoint method. Mathematics and Computers in Simulation 30, 405-418.
MassGIS. 1992. MassGIS datalayer descriptions and a guide to user services. MassGIS, EOEA Data Center, Boston, MA.
Meybeck, M. 1982. Carbon, nitrogen and phosphorus transport by world rivers. Amer. Jour. Sci. 282:401-450.
Miller, D. R., M. J. Focazio, M. A. Dickinson, and W. E. Archey. 1988. A User's Guide to a Model for Estimating the Hydrologic Effects of Land Use Change. Cooperative Extension Service, University of Connecticut, Storrs, CT, and the University of Massachusetts, Amherst, MA.
Monaghan, E. and A.E. Giblin. 1995. The effects of coupling between the oxic and anoxic layers of sediment on nutrient release to overlying water. Biological Bulletin 187:288-289.*
Morris, J.T. 1995. The salt and water balance of intertidal sediments: results from North Inlet, South Carolina. Estuaries 18:556-567.
Morris, J.T., and B. Haskin. 1990. A 5-yr record of aerial primary production and stand characteristics of Spartina alterniflora. Ecology 71:2209-2217.
Morris, J.T., B. Kjerfve and J.M. Dean. 1990. Dependence of estuarine productivity on anomalies in mean sea level. Limnol.Oceanogr. 35:926-930
Murphy, S.C. 1991. The ecology of estarine fishes in souther Maine high saltmarshes: access corridors and movement patterns. Dept. Fisheries and Wildlife Conservation, University of Mass. Amherst. 87 pp.
Newell, R. C. 1982. The energetics of detritus utilization in coastal lagoons and nearshore waters. Oceanologia Acta 1982:347-355.
Ney, J.J. (1993) Bioenergetics modeling today: Growing pains on the cutting edge. Transactions of the American Fisheries Society122, 736-748.
Nielson, L.P. 1992. Denitrification in sediment determined from nitrogen isotope pairing. FEMS Microbiology Ecology 86:357-362.
Nixon, S. and M. Pilson. 1984. Estuarine total system metabolism and organic exchange calculated from nutrient ratios: An example from Narragansett Bay. Pages 261-290 in: V. Kennedy (ed.) The Estuary as a Filter. Academic Press.
Nixon, S. W. 1981. Reminerilization and nutrient cycling in coastal marine ecosystems, pp. 111-138. In: B. Neilson and L. Cronin (eds.) Estuaries and nutrients. Humana, Clifton, NJ.
Nixon, S., C. Oviatt, J. Frithsen, B. Sullivan. 1986. Nutrients and the productivity of estuarine and coastal marine ecosystems. J. Limnol. Soc. S. Afr. 12:43-71.
Nixon, S.W., J.W. Ammerman, L.P. Atkinson, V.M. Berounsky, G.Billen, W.C. Boicourt, W.R. Boynton, T.M. Church, D.M. DiToro, R. Elmgren, J.H. Garber, A.E. Giblin, R.A. Jahnke, N.J. Owens, M.E.Q. Pilson, S.P. Seitzinger. 1996. The fate of nitrogen and phosphorus at the land-sea margin of the North Atlantic Ocean. Biogeochemistry 35: 141-180.
Norrman, B., U. Zweifel, C.S. Hopkinson and B. Fry. 1995. Production and utilization of dissolved organic carbon during an experimental bloom. Limnology and Oceanography 40:898-907.*
Olff, H., J.P. Bakker and L.F.M. Fresco. 1988. The effect of fluctuations in tidal inundation frequency on a salt-marsh vegetation. Vegetatio 78:13-19
Oviatt, C. A., J. G. Quinn, J. T. Maughan, J. T. Ellis, B. K. Sullivan, J. N. Gearing, P. J. Gearing, C. D. Hunt, P. A. Sampou and J. S. Latimer. 1987. Fate and effects of sewage sludge in the coastal marine environment: A mesocosm experiment. Mar. Ecol. Prog. Ser. 41:187-203.
Pennock, J., J. Sharp, W. Schroeder. 1994. What controls the expression of estuarine eutrophication? Case studies of nutrient enrichment in the Dleware Bay and Mobile Bay estuaries, USA. pp 139-146 in: Dyer and Orth (eds) Changes in fluxes to estuaries. Olsen and Olsen. 485 pp.
Peterson, B. J., B. Fry, M.A.J. Hullar, S. Saupe and R.T. Wright. 1994. The distribution and stable isotopic composition of dissolved organic carbon in estuaries. Estuaries 17:111-121.*
Peterson, B. J., M. Bahr and G. Kling (in press). A tracer investigation of nitrogen cycling in a pristine tundra river. Can. J. Fish. and Aquatic Sciences.
Peterson, B.J., R.W. Howarth and R.H. Garritt. 1985. Multiple stable isotopes used to trace the flow of organic matter in estuarine food webs. Science. 227: 1361-1363.
Portnoy, J. W. and A.E. Giblin. (1997). Biogeochemical effects of seawater restoration to diked salt marshes. Ecological Applications. (In press)*
Portnoy, J. W. and A.E. Giblin. (1997). Effects of historic tidal restrictions on salt marsh sediment chemistry. Biogeochemistry. 36:275-303.*
Quiros, R. and C. Baigun. 1985. Fish abundance related to organic matter in the Plata River Basin, South America. Transactions of the American Fisheries Society 114:377-387.
Reed, D.J. 1989. Patterns of sediment deposition in subsiding coastal salt marshes, Terrebonne Bay, Louisiana: The role of winter storms. Estuaries 12:222-227.
Rhee, G'Yull. 1972. Competition between an alga and its aquatic bacterium phosphate. Limnol. Oceanogr. 17:505-514.
Ricker, W. 1975. Computation and interpretation of Biological Statistics of Fish populations. Bulletin 191 Fisheries Research Board of Canada. Ottawa. 382 p.
Rigler, F. 1956. A tracer study of the phosphorus cycle in lake water. Ecology 37:550-562.
Rosenfeld, J.K. 1981. Nitrogen diagenesis in Long Island Sound sediments. Amer. J. Sci. 281:346-462.
Rountree, R., K. Able. 1993. Diel variation in decapod crustacean and fish assemblages in New Jersey polyhaline marsh creeks Estuar. Coast. Shelf Sci., vol. 37: 181-201.
Rozas, L.P. 1995. Hydroperiod and its influence on nekton use of the salt marsh: a pulsing ecosystem. Estuaries 18:579-590
Rybczyk, J.M., Garson, G. and J.W. Day, Jr. 1996. Nutrient enrichment and decomposition in wetland ecosystems: models, analyses and effects. Current Topics in Wetland Biogeochemistry. 2: 52-72.
Ryther, J.H. and W.M. Dunstan. 1971. Nitrogen, phosphorus, and eutrophication in the coastal marine environment. Science 171: 1008-1013.
Sammels, E. A. 1967. Water Resources of the Parker and Rowley River Basins, Massachusetts. USGS Hydrologic Investigation Atlas HA-247.
Secor, D., J Dean and S. Campana (eds). 1995. Recent developments in fish otolith research. Univ. South Carolina. 735 p.
Seitzinger, S.P. 1988. Denitrification in freshwater and coastal marine ecosystems: Ecological and geochemical significance. Limnol. Oceanogr. 33:702-724.
Seitzinger, S.P. and A.E. Giblin. 1996. Estimating denitrification in North Atlantic continental shelf sediments. Biogeochemistry 35:235-260.*
Seitzinger, S.P., W. Gardner and A. Spratt. 1991. The effect of salinity on ammonium adsorption in aquatic sediments: Implications for benthic nutrient recycling. Estuaries 14:167-174.
Sholkovitz, E. R. 1976. Flocculation of dissolved organic and inorganic matter during the mixing of river water and seawater. Geochim. Cosmochim. Acta 40:831-845.
Smaal. A. C. and T.C. Prins. 1993. The uptake of organic matter and the release of inorganic nutrients by bivalve suspension feeder beds. pp 273-298 in: R. Dame (ed.) Bivalve filter feeders in Estuarine and coastal ecosystem processes. Springer Verlag, Heidel burg.
Smith, S. V. and F. MacKenzie. 1987. The ocean as a net heterotrophic system: Implications from the carbon biogeochemical cycle. Global Biogeochemical Cycles 1:187-198.
Soil Conservation Service. 1986. Urban hydrology in small watersheds. SCS Technical Release 55. Washington, D.C.
Sullivan, B., P. Doering, C. Oviatt, A. Keller and J. Frithsen. 1991. Interactions with the benthos alter food web structure in coastal waters. Can. J. Fish. Aquat. Sci. 48:2276-2284.
Sutcliffe, W. H. Jr. 1972. Some relations of land drainage, nutrients, and particulate material and fish catch in two eastern Canadian bays. J. Fish. Res. Bd. Can. 29:357-362.
Thayer, G. W. 1974. Identity and regulation of nutrients limiting phytoplankton production in the shallow estuaries near Beaufort, NC. Oecologia (Berl.) 14:75-92.
Toolan, T., J. D. Wehr and S. Findlay. 1991. Inorganic phosphorus stimulation of bacterioplankton production in a meso-eutrophic lake. Appl. Environ. Microbiol. 57:2074-2078.
Tupas, L. and I. Koike. 1990. Amino acid and ammonium utilization by heterotrophic marine bacteria grown in enriched seawater. Limnol. Oceanogr. 35:1145-1155.
Uhlenhopp, A. G., J.E. Hobbie and J.J. Vallino. 1995. Effects of land use on the degradability of dissolved organic matter in three watersheds of the Plum Island Sound estuary. Biol. Bull. 189:256-257.*
Vallino, J.J. A bioenergetic approach to modeling microbial food webs. Proceedings of the NATO workshop on ecosystem modeling for the Black Sea. In Review.*
Vallino, J.J. and C.S. Hopkinson. 1997. Estimation of dispersion and characteristics mixing times in the Plum Island Sound Estuary. Estuarine, Coastal and Shelf Sci. (accepted).*
Vallino, J.J., and G. Stephanopoulos. 1993. Metabolic flux distributions in Corynebacterium glutamicum during growth and lysine production. Biotechnology and Bioengineering 41:633-646.*
Vallino, J.J., C.S. Hopkinson, and J.E. Hobbie. 1996. Modeling bacterial utilization of dissolved organic matter: Optimization replaces Monod growth kinetics. Limnol. Oceanogr. 41:1591-1609.*
Vezina, A. F. 1989. Construction of flow networks using inverse methods. Pages 62-81 in: F. Wulff, J. G. Field and K. H. Mann (eds.), Network Analysis in Marine Ecology. Springer-Verlag, Berlin.
Vorosmarty, C.J. and T.C. Loder,III. 1994. Spring-neap tidal contrasts and nutrient dynamics in a marsh-dominated estuary. Estuaries 17:537-551
Wagner, B. J. and J. W. Harvey (in press). Experimental design for estimating parameters of rate-limiting mass transfer: analysis of stream tracer studies. Water Resources Research.
Wainright, S. C. and C. S. Hopkinson. 1997. Effects of marine sediment resuspension on organic matter processing in coastal environments: a simulation model. J. Marine Systems. (in press).*
Warwick, J.J. (1989) Interplay between parameter uncertainty and model aggregation error. Wat. Res. Bull. 25, 275-283.
Webster, J., J. Meyer, P. Mulholland and B. Peterson. 1995. N uptake, retention and cycling in stream ecosystems: an intersite N-15 tracer experiment. NSF Proposal to DEB.
Welcomme, R. L. 1985. River fisheries. FAO Fishery Resources and Environment Division. Tech. Pap. 262:330 pp.
Weston, N.B., S.S. Carini, A.E. Giblin, G.T. Banta, C.S. Hopkinson and J. Tucker. 1996. Estimating denitrification in sediments of the Parker River estuary, Massachusetts. Biological Bulletin 191:334-335.*
Wheeler, P. A. and D. L. Kirchman. 1986. Utilization of inorganic and organic nitrogen by bacteria in marine systems. Limnol. Oceanogr. 31(5):998-1009.
Wikner, J., J.J. Vallino, G. Steward, D.C. Smith and F. Azam. 1993. Nucleic acids from the host bacterium as a major source of nucleotides for three marine bacteriophages. FEMS Microbiology Ecology 12:237-248.*
Willemsen, J. 1980. Fishery-aspects of eutrophication. Hydrobiol. Bull. 14:12-21.
Wollast, R. and G. Billen. 1981. The fate of terrestrial organic carbon in the coastal area, pp. 331-359. In: Flux of Organic Carbon by Rivers to the Oceans. Report of the workshop, Carbon Dioxide Effects Research and Assessment Program. U. S. DOE CONF-8009140.
Zhou, M. and Huntley, M.E. (1996) The principle of biological attraction, demonstrated by the bio-continuum theory of zooplankton patch dynamics. J. Mar. Res. 54, 1017-1037.
Zwolsman, J. J. G. 1994. Seasonal variability and biogeochemistry
of phosphorus in the Scheldt Estuary, South-west Netherlands. Estuarine,
Coastal and Shelf Science 39: 227-248.
RESEARCH MANAGEMENT AND SCHECULE
Overall direction and management is provided by Charles Hopkinson. Hopkinson continues in this role from the current LMER project. Research direction, strategic scheduling of major initiatives and budgetary matters are discussed collegially and decisions reached by consensus.
Our proposed project has expanded in scope and personnel over the current LMER. The LMER has 8 PIs at 3 institutions, while our LTER proposal has 11 PIs at 5 institutions; 6 if the LMER PIs remain. Personnel participation has been determined by consensus.
PIs and other staff at MBL meet monthly for information discussion and planning sessions. Summaries of these meetings are distributed by e-mail to all personnel. We will plan an intensive 2-3 day workshop of all researchers each winter to synthesize results across disciplines and to plan the next years research. It is important to note that 6 of the 11 PIs have offices within 10 m of each other and Vorosmarty and Buchsbaum regularly visit MBL. Thus a high degree of communication is insured.
Coordination of research is organized around the six Research Questions:
| Question | Coordinator |
| 1) Watersheds | Vorosmarty |
| 2) Marshes | Morris |
| 3) Planktonic | Hobbie |
| 4) Benthic | Giblin |
| 5) Higher Trophic Levels | Deegan |
| 6) Synthesis & Modeling | Vallino |
Cross-cutting experiments will each have additional coordinators: 1) N-15 addition to the Ipswich River - Peterson, 2) Tidal Creek Fertilization - Deegan, 3) Direct Marsh Fertilization - Hopkinson, 4) Detritus Removal Experiment - Hopkinson, 5) Mesocosms - Vallino. Buchsbaum, who works in the Plum Island watershed, will maintain local experiments, as necessary. Dionne is the scientific coordinator at the Wells National Estuarine Research Reserve while Allen is director of the Baruch Field Lab and North Inlet National Estuarine Research Reserve.
RESEARCH SCHEDULE
Year 1 - Long-term studies: Set up weather station and ISCO samplers for subcatchment streams. Begin the long-term measurements outlined on Table 1 except for the measurements of sediment accretion and marsh water table elevation. Primary production measurement s at fertilized marsh sites will be to collect pre-fertilization data. Questions & Experiments: Survey marsh and identify sites for long-term experiments. Survey marsh elevations for hydrodynamic mod3eling. Modeling: Expand the current adv3ection and dispersion model (AD) for spring-neap tides. Begin work on a tidally explicit hydrodynamic model. Outreach: We will continue to meet with several land and conservation groups including the Essex County Greenbelt Association and the Technical Advisory Committee of the Ipswich River Watershed Association. In the past we have also met occasionally with the Massachusetts Division of Environmental Protection, Division of Marine Fisheries, U.S. Fish and Wildlife Service and Coastal Zone Management to share our findings. We will institute a more formal set of meetings with these groups that also includes the scientific and management staffs from Wells and South Carolina. International: We will continue to participate in an international project lead by Ken Tenore to aid Portuguese scientists in develop a land-margin ecosystems project. Hopkinson and Deegan will both go to Portugal in the summer of 1997. We will continue to work on outreach and international collaboration as the opportunities arise (see Outreach Section).
Year 2 - Long-term studies: Continue all measurements outlined in Table 1. Begin measurements of sediment accretion and marsh water table elevation. Question & Experiments: Continue monitoring all marsh fertilization and detrital removal sites. Install SET tables, marker horizons and pressure transducers at control and experimental marsh sites. Begin growth and mortality experiments with fish. Modeling: Continue work on AD and hydrodynamic models. Begin work on existing estuarine food web (EFW) and general estuarine metabolism (GEM) model. Collaborations: Begin to apply the AD and GEM model to the Merrimack in collaboration with Driscoll. Compare estimate of N load from our watershed model to the Kremer N-Load model.
Year 3 - Long-term studies: Continue measurements (Table 1). Questions & Experiments: Carry out the whole river 15N addition experiment in the Ipswich River. Begin fertilizer additions. Begin marsh access by fish with flume work. Modeling: Complete AD and hydrodynamic models. Link hydrodynamic model to river routing model using results of tracer studies. Continue work on EFW and GEM. Collaborations: Complete the application of our AD and simplified GEM model to the Merrimack River.
Year 4 - Long-term studies: Continue measurements (Table 1). Questions & Experiments: Experiments on the pelagic systems using carboys and microcosms. Modeling: Incorporate data obtained in carboy and microcosm experiments into EFW and GEM. Begin work on spatially explicit fish production model linked to the hydrodynamic model.
Year 5 - Long-term studies: Continue measurements (Table 1). Questions & Experiments: Sediment microcosm experiment. Modeling: Incorporate results of sediment microcosm experiment into EFW and GEM. Extend fisheries model to include marsh flooding. Collaborations: Work with Vorosmarty to scale up GEM to Gulf of Maine.
Year 6 - Long-term studies: Continue measurements
(Table 1). Questions & Experiments: Large scale mesocosm experiment
using pelagic and benthic-pelagic mesocosms (Q3, 4 and 5). Modeling:
Use mesocosm data to test and refine EFW and GEM models. Complete fisheries
model. Collaborations: Work with Vorosmarty to scale up GEM to Gulf
of Maine.
DATA AND INFORMATION MANAGEMENT
Information Management
The objective of the Plum Island Sound Comparative Ecosystem Study (PISCES) data and information system is to provide a centralized network of information and data related to the Plum Island Sound Estuarine Ecosystem. This centralized network provides researchers associated with PISCES access to common information and data in addition to centralized long term storage. Researchers associated with PISCES are committed to the integrity of the information and databases resulting from the research.
PISCES information and databases are stored on a Silicon Graphics network server which is backed up on 8 mm tape nightly. Public access to PISCES data and information for the scientific community at large is provided through the PISCES World Wide Web home page on the Internet at the following URL: http://www.mbl.edu/plumisla.html
The PISCES LMER home page has been active since late 1995 and consists of a variety of information including personnel, data and published and unpublished papers and reports. The data section is broken down into eight sections consisting of maps, climate, watershed, estuarine physics, experiments, higher trophic levels, metabolism and nutrient chemistry, and models. The home page is updated annually and when a particular database update affects data already on the web.
Data Management and Coordination of Research Projects
Individual researchers are responsible for each of the six core research themes outlined in the PISCES LTER (Watersheds, Marshes, Planktonic Food Web, Benthos, Higher Trophic Levels and Modeling). Each researcher will communicate with the data manager regarding the design of the specific research project and subsequent incorporation of data and information into the PISCES database. Robert Garritt, a senior research assistant with The Ecosystems Center, MBL, has been database manager for the PISCES LMER and will continue the responsibility for overseeing the overall integrity of the data and information system for a PISCES LTER.
Contributions of Data to Database
Researchers on the PISCES LTER are expected to follow the guidelines as defined in the LTER Guidelines for Site Data Management (Porter and Callahan 1994). The current PISCES and North Inlet, SC databases follow the LTER guidelines and we plan on developing similar database management with Wells Bay, ME in the future. Research conducted using the facilities of the PISCES LTER will be expected to comply with the following policy:
4) An electronic link will be established between remote site databases (North Inlet-NERR, Wells Bay-NERR) and PISCES for accessibility of long term data sets.
Researchers on the PISCES LMER/LTER have been and will continue to be encouraged to both publish and contribute data to the PISCES database. It is recognized that investigators on PISCES have first opportunity for use of data in publications but there is also the realization for timely submittal of data sets for incorporation into the PISCES database. Four categories of data are recognized as outlined in the LTER Guidelines for Site Data Management (Porter and Callahan 1994). PISCES specific policy for data sharing is as follows:
Data Type I. Published data and meta-data (data about data)
Policy: Data are available upon request without review.
Data Type II. Collective data of the LTER site (usually routine measurements generated by technical staff.
Policy: Data are available for specific scientific purposes within one year after generation.
Data Type III.Original measurements by individual researchers (experimental data).
Policy: Data are available for scientific purposes two years after generation. Data can be released earlier with permission of the researcher.
Data Type IV.Unusual long-term data collected by individual researchers.
Policy: Data are available with permission of the researcher.
Future Objectives
Data sets are currently in spreadsheet and text format which has been adequate in the past due to the relatively small size of the data sets. We anticipate the need for converting data sets of routinely measured parameters to spatially referenced databases which will allow compatibility of format for use in Geographic Information Systems (GIS). We also plan to incorporate a query feature for seeking out subsets of the databases.